
The ashwagandha leaf "ban" story has a twist. Some articles were pulled, government documents went offline, and the science cited doesn't support what regulators claimed.
Two documents, issued one day apart by two different Indian government agencies, have touched off a global debate about what ashwagandha is, which part of the plant belongs in your supplement, and whether the trade press' characterization of a regulatory action reflects what those documents actually say.
India controls the overwhelming majority of the world's ashwagandha supply, so when its government acts on the ingredient, the industry pays attention. When word spread in late April 2026 that India had "banned" ashwagandha leaves, brands started asking questions and some suppliers declared victory through paid media.
Two Documents, One Story Being Told Many Ways
The reality is more complicated. One of those documents (the one from the Ministry of Ayush) does use binding language. The other, from FSSAI, is literally titled "Advisory" and reminds food business operators that existing 2016 regulations already restricted the ingredient to the root in food products in India.
We investigated, and along the way, found that articles were removed, posts were edited, and previously-published advisories and safety documents completely disappeared from government websites. What began as an advisory is turning into a story of what happens when regulators and industry media push the boundaries just a bit too far on behalf of their sponsors.
Calling both documents a ban isn't wrong in a casual sense, at least not locally in India. But characterizing the package as a unified global ban with supply implications that extend beyond India's borders is a different claim. That's the one worth examining, and the whole story is covered in this article. But first, some necessary disclosure:
Full Disclosure: Sponsored Article
Arjuna Natural, the maker of the Shoden ashwagandha extract, is an ongoing partner of PricePlow's and sponsored this article and associated video. However, PricePlow's editorial position on supplement industry coverage goes beyond any single supplier relationship. Our primary concern is consumer safety, efficacy, and the health of the industry as a whole. This story impacts all three, and the public conversation has only been dominated by one perspective.
Before diving in, sign up for our alerts Arjuna Natural and ashwagandha so that you can get notified when there are updates to the situation:
Subscribe to PricePlow's Newsletter and Alerts on These Topics
What the Documents Actually Say
-
The Ayush Directive (15 April 2026)
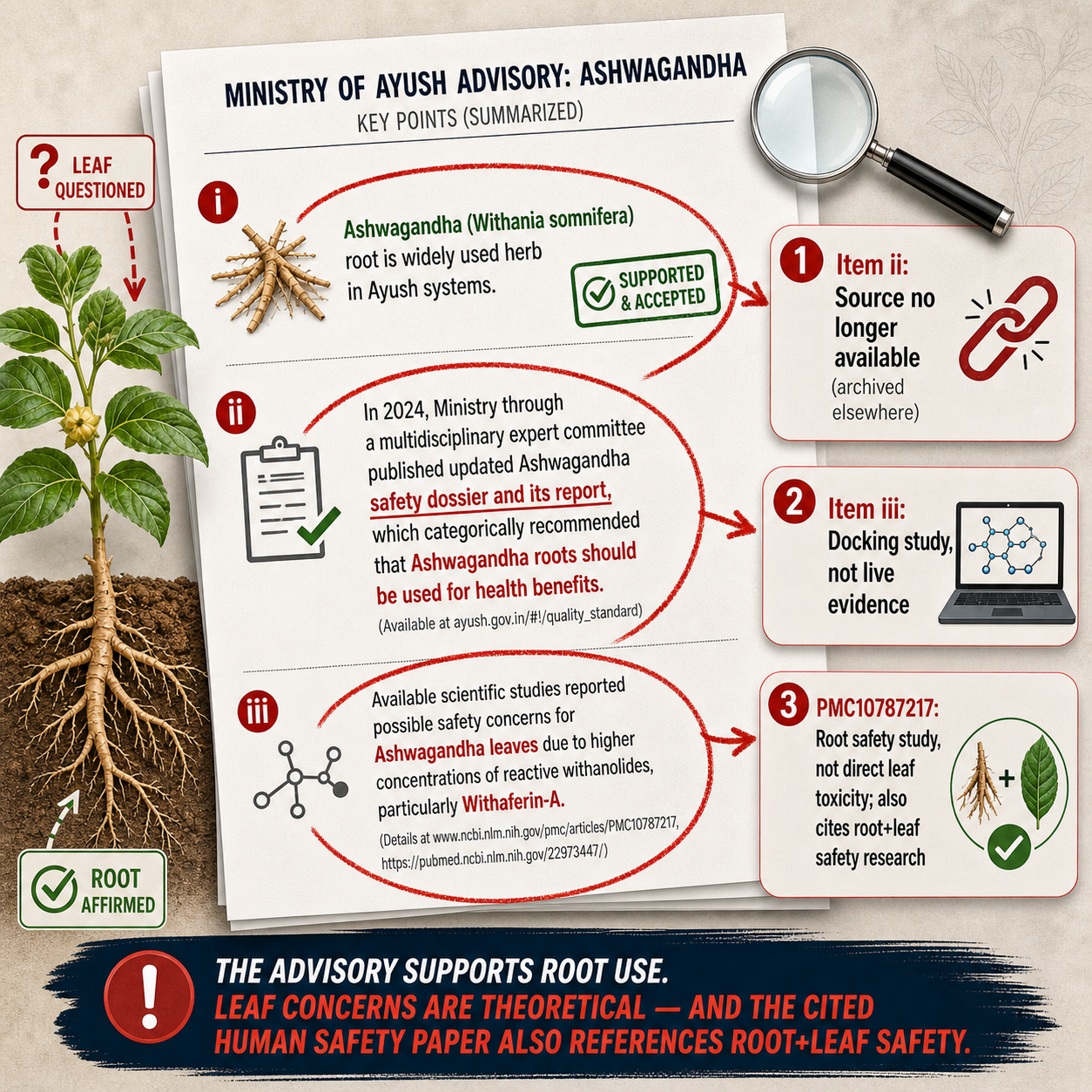
The Ministry of Ayush is India's government ministry responsible for traditional medical systems including ayurveda, yoga and naturopathy, homeopathy, and more. Their file reference T-13020/4/2022-DCC-Part(2), is the harder of the two documents. Addressed to all Ayush drug manufacturers, exporters, and sellers, its operative language is unambiguous: the use of ashwagandha leaves in crude, extract, or any other form in Ayush drugs and products "is strictly prohibited".[1]
The directive rests on three pillars:
- First, it references a 2024 safety dossier produced by a multidisciplinary expert committee, which concluded that ashwagandha root is the appropriate part for health use.[2]
- Second, it points to scientific studies on withaferin A safety concerns.
- Third, it invokes Rule 161 of the Drugs Rules, 1945, which requires plant parts used in products to be clearly labeled.
The directive was signed by Shiela Tirkey, Under Secretary to the Government of India.
-
The FSSAI Advisory (16 April 2026)
The FSSAI (Food Safety and Standards Authority of India) is India's national food regulator. Their document, file reference F. No. RCD-15001/11/2021-Regulatory-FSSAI, is a different animal. Its title is "Advisory", and not an order nor a prohibition.[1] The document advises all Food Business Operators to "ensure strict compliance" with Schedule IV of the existing 2016 Health Supplements/Nutraceuticals regulations, which already limited the ingredient to roots and root extracts.
FSSAI isn't creating a new rule. It's reminding operators that a rule from 2016 exists and will be enforced. The advisory warns that deviations "shall attract action" under the Food Safety and Standards Act, 2006. It was signed by Dr. Amit Sharma, Executive Director (Compliance Strategy).
-
A Note on Terminology
U.S.-based readers should resist importing FDA interpretive frameworks onto Indian regulatory language. An "advisory" from FSSAI may carry different practical weight than the word implies in an American context. The Ayush directive uses binding language. Whether the FSSAI advisory, rooted in existing 2016 regulations, constitutes an actual new ban or an enforcement reminder is a question to be answered, but it seems that nothing has changed for foods in India since 2016. Both documents apply only to products manufactured and marketed within India.
From 2021 to 2026: How We Got Here
This isn't the first time Ayush has taken aim at ashwagandha leaf. In October 2021, the ministry issued a document titled "ADVISORY" (file L.11011/9/2021-DCC) advising manufacturers not to use ashwagandha leaves in crude or extract form for therapeutic purposes.[3] The rationale at that point: leaves aren't cited in classical Ayurvedic texts as the preferred plant part, and there weren't sufficient independent safety and efficacy studies to justify their use.

Shoden brings a new era to ashwagandha supplements with 35% glycowithanolide standardization. Research shows it delivers powerful effects at just 60-120mg daily, compared to 600mg+ with traditional extracts.
Industry pushed back. Manufacturers, exporters, and trade associations made representations arguing the advisory was scientifically premature. In November 2021, Ayush responded by forming an Expert Group to re-examine the question, citing discussions with stakeholders.[4] That group's work eventually produced the 2024 safety dossier now cited as the scientific foundation of the April 2026 directive.
Stemming from a 2020 Danish Risk Assessment
The 2024 dossier deserves a careful read. It wasn't originally framed as an anti-leaf document. Its primary purpose was to defend ashwagandha root against the Danish Technical University (DTU) Food Institute's 2020 risk assessment, which raised concerns about the ingredient's safety and contributed to regulatory restrictions across several European countries.[2]][5] In building that defense, the committee reviewed phytochemical concentration data across raw plant parts and concluded that root use was preferred and appropriate. The dossier includes comparative data showing withaferin A at roughly 0.92mg/g in roots and roughly 22.31mg/g in aerial parts.[2]]
That figure sits at the center of the current scientific dispute.
The April 2026 directive also signals enforcement frustration: it notes that "certain Ayush manufacturers are still using Ashwagandha leaves" despite the 2021 advisory, suggesting the shift from advisory to binding directive was at least partly a response to ongoing non-compliance.
Two Documents No Longer Live Online?
Interestingly, the 2021 Advisory and the 2024 safety dossier have both been taken offline, despite having recently been online. Their archive links are fortunately still up:
- 2021 Advisory:
- 2024 Safety Dossier:
While some regulating authorities (such as the FDA) have automatic archive periods, the takedown of the recent safety dossier -- which was online at the beginning of our investigation -- raises a great deal of suspicion.
“Ban” or “Advisory”? Why the Word Matters
The Ayush directive uses binding language. For products classified as Ayush drugs and sold within India, leaf use is not permitted. Calling that a ban is accurate.

Arjuna Natural's Shoden-R ashwagandha just won Ingredient Idol at SupplySide Global 2025. The root-only extract is water-soluble, works at 60mg, and finally makes ashwagandha viable in beverages, gummies, and RTD formats.
The FSSAI document is titled "Advisory" and enforces regulations already in force since 2016. Whether it constitutes a new ban or an enforcement clarification depends on how you're counting, and on whether you believe those 2016 regulations were being enforced before April 2026.
Soon after the April 2026 documents were issued, Ixoreal Biomed, the creators of KSM-66® ashwagandha root extract, posted to LinkedIn. The current version of that post describes the company's foundational choice to use root alone and states that Indian regulators "prohibited Ashwagandha leaves, whether crude or as extract, across both Ayush and food products."[6] Note the word "ban" being used in the URL, but no longer anywhere in the post, which signals some of the potential verbiage in the pre-edited post.
Banned in Only India? Can Purified Leaves be Exported?
In trade press coverage, Kartikeya Baldwa, CEO of Ixoreal Biomed, described the directives as a market correction that aligned with millennia of traditional Ayurvedic use. He argued that when India defines what ashwagandha must be at the point of origin, that standard shapes raw materials exported to finished product markets globally, and that the root-only standard now applies beyond India's domestic market.[7]
Whether that legal interpretation holds is a question that must be clarified, because these documents do not explicitly state that. Arjuna Natural's customer statement sent to their Shoden customers takes the opposite position, noting that the directives "apply to products manufactured, marketed, and sold within India" and don't, on their own, alter the regulatory status of ashwagandha root and leaf extracts in export markets.
The framing question: does describing both documents together as a blanket ban, then asserting that standard travels globally, accurately represent what those documents say?
The Withaferin A Question
Withaferin A is a withanolide present in both the root and aerial parts of ashwagandha. It's not a contaminant -- it's one of the plant's primary bioactive compounds, and it does appear at meaningfully higher concentrations in raw leaf and aerial parts than in raw root. The 24-fold ratio in the 2024 dossier isn't seriously disputed.[2]
But the scientific question isn't just how much is in the leaf. It's whether concentrations in oral supplements produce harm at the doses people actually take.
The Studies Behind the Directive
The Ayush directive references two papers to support its withaferin A safety concerns.[1] Understanding what those papers actually studied matters for evaluating the directive's scientific basis.
- The first is Vaishnavi et al. (2012), a molecular docking study, meaning it's a computational simulation predicting how compounds might bind to target proteins![8] Molecular docking generates hypotheses for further laboratory testing, but it can't demonstrate actual cytotoxicity or any biological effect in cells.
So this is not even an in-vitro experiment, and it's not a safety evaluation of oral ashwagandha consumption.
- The second reference is Kalaivani et al. (2023) in Toxicology Reports, funded by Shri Kartikeya Pharma -- the manufacturer of KSM-66.[9] That study evaluates the acute and sub-chronic safety of a standardized aqueous root extract, not a leaf-containing product. Its conclusions support root safety... but they don't demonstrate leaf toxicity.
But it gets more interesting. In the discussion, the paper benchmarks its KSM-66 root extract against Antony et al. (2018) as the prior safety reference, writing that its results are "consistent with previous study by Antony et al." The Antony et al. study, however, tested Shoden: a root-and-leaf extract![10] So the same paper that warns about leaf toxicity in its introduction then validates its own root extract's safety by citing a root-and-leaf study. Both claims can't be true.
To use this study as evidence against leaves effectively commits fallacy of the inverse (also called denying the antecedent). The regulatory directive's supporting literature doesn't hold up under actual scrutiny.
Ultimately, in vitro cytotoxicity in cancer cell lines at high concentrations of isolated compounds doesn't automatically translate to toxicity at oral supplementation doses. That's a standard scientific caution... the directive's supporting literature simply isn't thorough enough, especially when it flies in the face of other research that was not cited:
What Broader Research Shows
The broader safety literature on root-and-leaf extracts tells a different story.
Kerry's aqueous root-and-leaf Sensoril extract is the subject of a 2026 OECD-compliant 90-day rat study published in Phytotherapy Research. Researchers found no toxicologically relevant changes at up to 4,000mg/kg/day, roughly 19 times the standard human dose, with full genotoxicity clearance across three OECD-compliant tests.[11]
Arjuna reports preclinical data showing a NOAEL of 1,000mg/kg/day in a 90-day rat study and an acute LD50 exceeding 2,000mg/kg, with normal findings across all parameters. A 90-day human safety study at 480mg/day in 62 participants is currently under peer review.[10]
Dr. Thomas Brendler of Plantaphile has been leading a team reviewing close to 140 clinical trials on ashwagandha for a special issue of Phytotherapy Research. His team found insufficient evidence to establish causality for adverse events, regardless of plant part. In NutraIngredients, Brendler described the action as "disproportionate, unless it were a politically motivated trade-off to dissuade EU regulators from further scrutiny."[7]
The Pharmacovigilance Picture
The adverse event data deserves an honest read, without weaponizing it for either side.
The Netherlands Pharmacovigilance Centre Lareb has received reports of hepatic adverse events associated with ashwagandha products. Those reports involve products derived from both leaf-containing and root-only preparations.[12-15] Every active herb carries some risk profile. The liver processes nearly everything that enters the body, and consumers with existing hepatic conditions warrant caution regardless of plant part. This isn't an argument against the regulation. It's an argument for applying scientific rigor to all ashwagandha safety questions equally, not just leaf-specific ones.
Interestingly, Netherlands' own reports mention "root" more than they do "leaf".[13,15] Additionally, adverse events have been reported in various industry-funded root-only studies,[16-19] though they're often minor, mild, and transient. There's likely more to the entire safety story, and we need to look more closely at the exact constituency and standardization of all extracts on the market.
The Coalition’s Response and Process Concerns

Bennito Russo, Keely Johnson, and Nipen Lavingia from Arjuna Natural's team reveals the science behind Shoden ashwagandha's superior bioavailability and their industry-leading botanical standardization on the PricePlow Podcast #171.
A coalition of approximately 16 manufacturers, including Sabinsa, Kerry, and Arjuna Natural, has reportedly gathered in Delhi to coordinate a response to the directives. Legal action is reportedly under consideration, though a public timeline hasn't been confirmed.[7]
-
The Scientific Argument
Shaheen Majeed, global CEO of Sabinsa, argues that the directive misread the science. His position: if regulators had genuine concerns about withaferin A concentrations, the appropriate response would have been establishing acceptable limits for dietary supplement use, not a blanket prohibition. Majeed also frames withaferin A as one of the more promising compounds in cancer biology, noting that restricting access to it categorically is a loss for science.[7]
Dr. Mukesh Summan, former FDA toxicologist and global director of toxicology at Kerry, is actively engaging with Indian regulators to seek reconsideration, citing classical Ayurvedic citations for leaf use and arguing the directive doesn't fully reflect the weight of scientific evidence.[20]
Nipen Lavingia, brand innovation advisor at Arjuna Natural, describes Shoden's root-and-leaf combination as the product of bioactivity-guided fractionation: a structured process that begins with whole-plant phytochemical mapping and activity screening across biological models before extraction is optimized. The combined extract produces a standardized withanolide glycoside profile that the root alone can't replicate, and it's the material on which Shoden's clinical evidence base was built.[7]
-
The Procedural Questions
Coalition members have raised concerns about how the FSSAI advisory was developed. Industry sources allege the process didn't include consultations typically expected before a restriction of this scope, including no hearings with relevant scientific advisory bodies, the herbal industry, farmers, or trade associations. They also note the document was signed by an official below the executive head level.
These are claims from coalition members, but the FSSAI should consider clarifying its standard process for advisory issuance.
-
The Stakes
Majeed offered the clearest picture of what the directive affects economically. Approximately 25,000 acres in India are dedicated to ashwagandha cultivation annually, producing roughly 5,000 metric tons of roots and 7,500 metric tons of aerial parts. Across the full supply chain, 250,000 to 300,000 people depend on the crop.[7] Leaf material that previously generated farm income now has no domestic market under the new rules, and the safety demonstrated in Shoden-based studies indicates that their refinement process is high-quality. You can learn more about their standardization for withanolide glycosides in a previous sponsored article, "Shoden and the Ashwagandha Evolution: The Rise of Glycoside-Enhanced Extracts".
-
On the Topic of Removed Papers and Edited Articles
Jessie Cavanaugh of Nutraceuticals Research Institute and Keely Johnson of Arjuna Natural break down the science of cortisol awakening response, ashwagandha research methodology, and Shoden's groundbreaking three-arm study on Episode #207 of the PricePlow Podcast.
To top it all off, a paid advertorial regarding the matter was posted to NutraIngredients.com on April 27, 2026 but the link came up dead on April 29th at 2:00 PM Central Time. It is once again live, currently showing an update date of April 27, 2026.[21] What was changed in those edits? There are unfortunately no public archives of the original article. We've reached out to NutraIngredients for comment and will update this if there are any.
In response to the entire matter, Blake Ebersole of the Ashwagandha Standards Alliance took aim at how the industry narrative is being shaped. The Alliance supports enforcement against undisclosed substitution and valid labeling. It rejects using paid media to "convert a commercially useful false interpretation into industry-wide consensus".[22] Ebersole's post also drew a line that the broader debate tends to blur: undisclosed substitution of leaf for root is a genuine adulteration issue, while disclosed root-and-leaf products are a different category entirely.
What This Means for U.S. Formulators and Consumers
-
Regulatory Status Outside India Is Unchanged
The Indian directives don't change the legal status of ashwagandha in the U.S. or Canada. Under DSHEA, Withania somnifera root and leaf extracts remain lawfully marketable as dietary supplement ingredients. Under Health Canada's NHPD framework, the same materials remain eligible under existing monograph and licensing requirements. Arjuna, Kerry, and Sabinsa have all confirmed uninterrupted production for export markets, according to Arjuna Natural's statement emailed to Shoden customers.
-
Questions Worth Asking
Rather than treating the Indian action as settled global safety guidance, formulators should focus on the operational questions that actually affect product quality and consumer trust:
Is the plant part clearly disclosed on the label? Is withanolide standardization measured by a validated, reproducible method (USP or British Pharmacopoeia standards) rather than a proprietary method that can't be independently audited? Does the supplier provide lot-level identity testing and traceability from cultivation through finished extract? Are the safety and efficacy studies conducted on the actual material being supplied, not a different plant-part ratio?
These questions matter regardless of where the regulatory debate lands.
-
The Adulteration Problem Is Real, and Separate
There's a legitimate concern embedded in the Indian regulatory action that deserves acknowledgment on its own terms.
The American Botanical Council's October 2025 Laboratory Guidance Document addresses the problem of ashwagandha aerial-part substitution into products labeled as root-only.[23] Undisclosed leaf in a root-labeled product isn't a plant-part preference dispute -- it's literally fraud. A 2025 study by Avula et al. in Phytotherapy Research found that current pharmacopeial methods don't reliably distinguish root from aerial-part blends, creating an analytical gap that complicates both enforcement and self-regulation.[24]
Addressing undisclosed substitution is legitimate regulatory work. But it's a different task from restricting disclosed, transparently labeled, safety-studied root-and-leaf products. Conflating those two issues makes it harder to solve either one.
For consumers: read the label and know the plant part. If it's not specified, ask. As a brief note on terminology, Shoden is a disclosed root-and-leaf extract with its clinical evidence built on that specific combination. Shoden-R is Arjuna's root-only option for formulators wanting that profile.
The Questions That Remain
Here's what's documented: two documents, two agencies, two different scopes, and a genuine scientific dispute over whether concentration data from in vitro cancer cell studies justifies restricting disclosed and safety-studied leaf-containing extracts.
Here's what's not yet resolved. Will the coalition's legal action proceed, and on what grounds? Will FSSAI respond publicly to the procedural concerns raised? Will the broader conversation about pharmacopeial methods for detecting root-and-leaf blends gain traction in regulatory science? And will India's domestic rule, in practice, reshape what reaches shelves in North America and Europe?
The deeper question this moment raises isn't about any specific plant part or any specific supplier. The ashwagandha category has needed better standardization, better labeling discipline, and a cleaner separation between adulteration enforcement and competitive positioning for years. This regulatory action could force that conversation forward, and we're here for that, because there does seem to be a small safety signal underlying even root extracts. Figuring it out and ensuring the safest extracts is the best path forward. Watching which way it goes is exactly what we're here for.
To stay current on this story as it develops, sign up below for Arjuna Natural news alerts -- this situation is evolving and we'll cover it as it does.






Comments and Discussion (Powered by the PricePlow Forum)