As a mineral nutrient found throughout the body, it's tough to dispute the importance of zinc in the diet. Extensively involved in the metabolism of proteins, lipids, nucleic acids, as well as proper gene transcription,[1] zinc plays a crucial role in optimal physical health.
Despite its importance, however, zinc can't be synthesized by the body, nor can it be stored in significant amounts. The majority of it is actually stored in muscle and bone tissue, and thus, not always readily available.[2] This is why you have to get zinc from food and supplements, and get it on a regular basis.
There is an abundance of well-established research demonstrating that adequate zinc levels are beneficial for immunity,[3-5] fertility,[6] cognition,[7] healing,[8] healthy hair and skin,[9] taste and smell,[10] and numerous other biological systems. The question is not whether zinc is beneficial, but instead, what type to use and how much you need. The amount depends on one's diet, age, and current health status, but the optimal type of supplemental zinc is often debated.
In this article, we present our recommendation for a highly bioavailable zinc option.
Moving on from zinc oxide
For decades, zinc oxide was utilized in multivitamins and mineral supplements, primarily due to its low cost, ease of procurement, and relatively low weight. But over the years, research has shown that zinc oxide has awfully low bioavailability.[11-13] One study even shows that zinc oxide has zero absorption in some people! As consumers have become savvier researchers, and dietary-supplement education continues to improve, the market has demanded more from supplement manufacturers who, in turn, have distanced themselves from zinc oxide in search of forms that provide higher bioavailability and absorption.

Meet Zinc Picolinate: A highly-bioavailable form of zinc powered by the incredible benefits of picolinic acid. You can find it in Nutrition21's Zinmax.
There are numerous types of zinc ingredients on the market, each with varying absorption strategies and profiles. While some forms show improvement by sporting higher bioavailability than zinc oxide, they do so by sacrificing flavor and aroma. These "new and improved" forms may be easier for the body to absorb, but only if you can stomach a hard-to-disguise bitter taste and pungent odor.
Introducing zinc picolinate: Meet Nutrition21's Zinmax
There is one form of high-bioavailability zinc that overrides these concerns, yet still doesn't get enough attention: zinc picolinate. Bound to picolinic acid, this is a unique and powerful way to supplement the critical mineral that utilizes the same mechanism found in our own digestive system. Zinmax is the patented form of zinc picolinate from Nutrition21, the sponsors of this article and creators of Chromax -- a well-known and highly-researched chromium picolinate ingredient that employs a similar delivery strategy.[14] Its molecular formula is Zn(C6H4O2N)2.
In this article, we detail an extraordinarily well-performed study that demonstrates zinc picolinate's significantly better bioavailability when compared to two alternative forms of supplemental zinc.[15] Before that, however, we dive into the research that led to that study, explaining the importance of the picolinic acid itself.
Our findings are below, but first, sign up for our Nutrition21 and Zinmax news alerts if you want to continue receiving information about the company's research-backed ingredient innovations:
Subscribe to PricePlow's Newsletter and Alerts on These Topics
This area is reserved for Team PricePlow's upcoming Ingredients video.
Subscribe to our channel and sign up for notifications so you catch it when it goes live!
We've covered chromium picolinate numerous times on this blog, but chromium isn't the only mineral that binds well with picolinic acid. To understand how the scientific community figured it out, a bit of backtracking through the research is helpful:
Zinc-binding ligands: the body's natural zinc absorption mechanism
In the 70s, scientists discovered zinc-binding substances in the gastrointestinal tracts of rats, known as low-molecular-weight zinc-binding ligands, or LMW-ZBL.[16,17] These substances, which included picolinate (picolinic acid) and citrate (citric acid), were identified as zinc-binding ligands (ZBLs).[18-20]
Similar ligands were seen in pancreatic secretions from dogs, leading researchers to theorize that these molecules facilitate the absorption of dietary zinc.[21] When fed a zinc-deficient diet, rats' zinc absorption percentage went up (along with the secretion of ZBLs) in an effort to compensate for falling zinc levels.
Additionally, ZBLs were also found in both human and rat milk,[22,23] leading scientists to believe that critical mechanisms were involved.
Picolinic acid: the best ZBL?
Identified as a metabolite of the tryptophan amino acid,[24] picolinic acid (C5H4N(CO2H)) is known as a bidentate chelating ligand that binds with zinc and other minerals such as chromium.[25] The best way to ensure the natural availability of picolinic acid is to eat a diet with enough complete essential amino acids (ensuring enough tryptophan intake) and to ensure enough bioactive vitamin B6 intake. Since kyureninase is one of the enzymes used in the pathway that converts tryptophan to picolinic acid, and kyureninase is dependent upon active vitamin B6,[26] proper dietary intake of both nutrients are required for optimal zinc-binding ligand production.
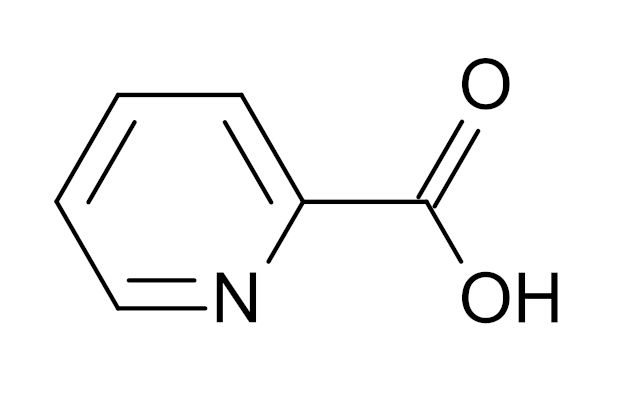
The incredible picolinic acid molecule, a zinc-binding ligand that helps the body absorb dietary zinc.
Mechanistically, research showed that picolinic acid boosts zinc metabolism through improved intestinal absorption.[27,28] Understanding this, researchers moved to animal studies where they demonstrated that picolinic acid successfully promotes the intestinal absorption of zinc in rats.[26,29]
This led researchers to wonder if binding zinc to citric acid or picolinic acid would yield benefits in humans— supporting the body's naturally-existing absorption mechanism.
Zinc Picolinate: A highly-bioavailable form of zinc
With zinc and ZBL research from the 1970s well-established, scientists decided to put them to the test in a controlled human study. The results were published in Agents and Actions in 1987.[15] The scientists recruited 15 healthy human subjects who had no zinc deficiencies but did have zinc intakes slightly below the recommended daily allowance. For 16 weeks the participants tested three different oral zinc supplements by rotating through four separate groups in varying sequences yielding 50 milligrams of elemental zinc each (or placebo):
Zinc picolinate gets a significantly greater amount of zinc into the hair,[15] which is a great way of determining that it's made its way through the body.
- Zinc picolinate - 144 milligrams
- Zinc citrate - 146 milligrams
- Zinc gluconate - 385 milligrams
- Placebo - 0 milligrams
Tablets were taken three times daily, midway between dinner and bedtime. After each four week period, there was a two week washout where no supplements were taken.
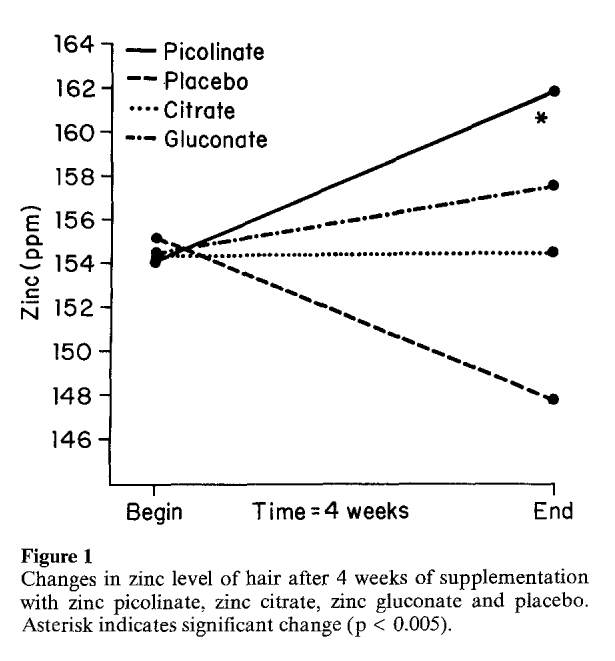
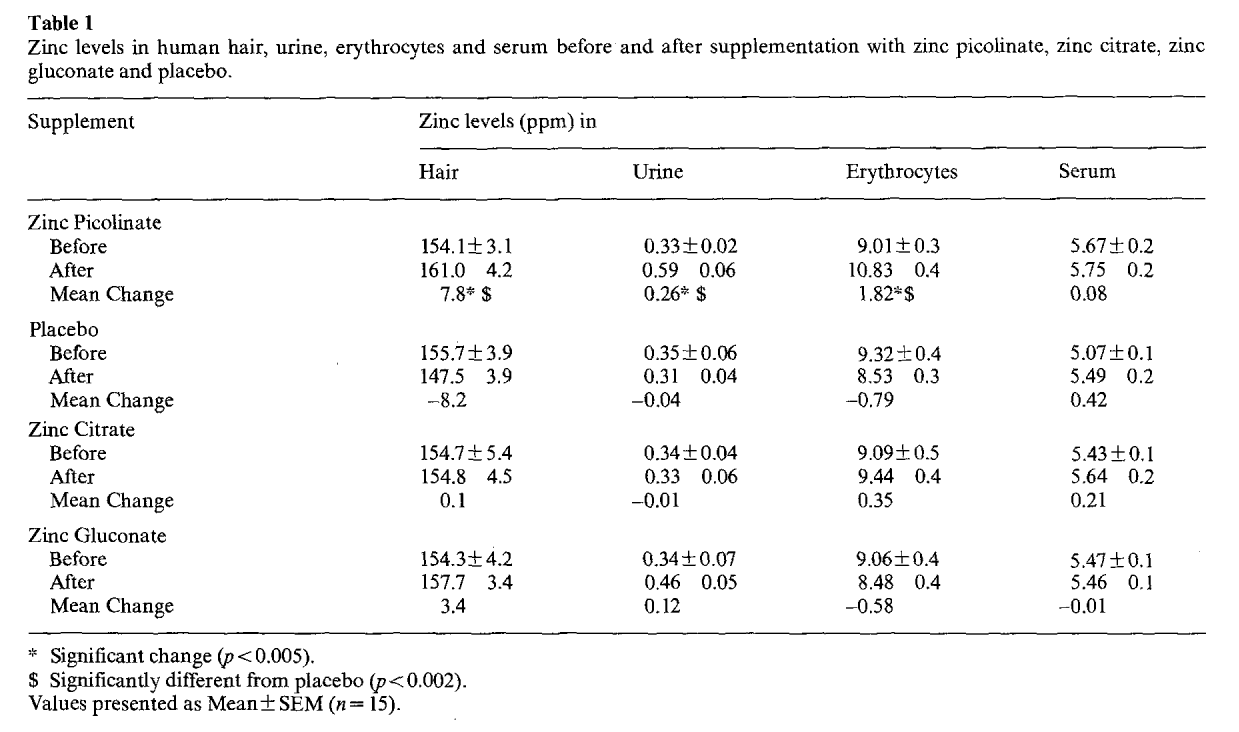
On the days before and after each four week period, the researchers measured zinc content in the participant's hair, urine, erythrocytes (red blood cells), and serum. The analysis determined that only zinc picolinate raised zinc levels to a statistically significant degree.[15]
Interestingly, only zinc picolinate showed increases in hair, urine, and red blood cells. Although zinc gluconate and zinc citrate demonstrated greater improvements in serum zinc levels, in other areas, their results were sporadic. For instance, the zinc gluconate groups showed lower zinc levels in red blood cells, while zinc citrate showed slightly lower zinc urine levels and barely different hair levels.
Another interesting point is that the placebo groups had non-significantly increased zinc serum levels.[15] This could have likely been due to chance, but the entirety of the data above leads us to conclude that zinc needs a high-quality transport system, and picolinic acid works the best across multiple boards.
Additionally, this research shows the importance of collecting multiple data points -- serum levels may not be indicative of what's actually getting into red blood cells and where it's needed.
Other specific zinc picolinate studies
Zinc picolinate has been studied multiple times in terms of improving taste sensation,[30,31] succeeding where other forms of zinc sulfate and zinc gluconate had previously failed to show overall significant effects.[32,33] In the zinc picolinate studies, the subjects had significantly improved serum zinc levels compared to placebo.[30,31]
Zinc as an antioxidant

Zinc exerts its benefits through a variety of ways, from structural support to antioxidant support to beneficial chemical displacement, leading to systemic health improvements
Other recent research has demonstrated antioxidant-like effects from zinc, yielding beneficial effects on oxidant-antioxidant balance.[34] It's suggested that zinc functions as an antioxidant in four ways:[35-37]
- Zinc competes with iron and copper, displacing them and making them available to bind to ferritin.
- Zinc is a cofactor for enzymes that are critical for a high-functioning antioxidant defense system.
- Zinc physically protects cells against oxidative damage and stabilizes membranes.
- Zinc binds to sulfhydryl groups in proteins, protecting them from oxidation.
These effects aren't exclusive to zinc picolinate, but zinc picolinate shows quite a significant boost in antioxidant activity alongside zinc levels,[34] consistent with every other study we've seen performed on this complex of the element.
Research has also demonstrated that zinc picolinate has antiviral effects,[38] which is also in line with other forms of zinc.
Safety and tolerable upper limits
Zinc picolinate has been approved by the European Food Safety Authority (EFSA) as a supplemental mineral source that is safe for adults and children.[39]
As for dosage, zinc picolinate should be treated similarly to other forms of zinc: look at the amount of elemental zinc provided, and compare it to the recommended daily allowance. Zinc supplements will generally provide anywhere from 25 to 50 milligrams of elemental zinc, but it's worth noting that 40 milligrams is the United States government's recommended upper intake level,[40] even though 50 milligrams has been safely studied.[15]
Additional articles mentioning Zinmax on The PricePlow Blog will be highlighted below:
Modern diets strike again: do we need help making picolinic acid?

For over 25 years, Chromax chromium picolinate has been improving insulin sensitivity. We argue that it's only gotten better, as dietary deficiencies have gotten worse over this time period.
Upon researching the zinc-binding ligands (ZBLs) and understanding the criticality of picolinic acid, we come to realize that ingesting zinc picolinate isn't only useful for its ability to provide zinc, it's also helpful as a source of picolinic acid! In order to create this incredibly powerful binding agent, the body requires both tryptophan and active vitamin B6[26] -- an amino acid and a B-vitamin that some dieters may not get enough of these days, especially if they're not eating enough meat.
We posit this: zinc picolinate (ZinMax) and chromium picolinate (Chromax) supplements aren't just useful for the respective zinc and chromium that they provide -- they're also beneficial because they offer additional picolinic acid needed to bind to these (and other) minerals!
Nearly every time we dive deep into an area of nutritional research, we find ourselves staring at the importance of consuming whole food, high-protein diets — and the research has been around for half a century at this point to confirm these underpinnings.
Zinmax: A high-bioavailability zinc picolinate to consider
Numerous zinc-based supplements have come out in the past couple of years, with some fairing better than others. However, after considering the above research on picolinic acid, we believe that the dietary supplement community may collectively be missing a major piece of the puzzle.
Zinc picolinate provides a transport mechanism that's naturally found in the body. Yet, the same dietary concerns that cause low zinc levels may also cause low transport capabilities. By binding zinc to its "native" transporter, Zinmax uses a novel approach to increasing zinc bioavailability[14] — and it has been shown to work beyond mere plasma increases.
In recent years, Nutrition21 has innovated in the sports nutrition industry numerous times, but their expertise in chromium picolinate (Chromax) reminds us that their ingredients have extremely wide-ranging applications. Chromium picolinate's success can be attributed to the improved uptake thanks to picolinic acid -- so it naturally makes sense to consider the same for zinc with zinc picolinate.





Comments and Discussion (Powered by the PricePlow Forum)