
NNB Nutrition's GlucoVantage® targets the Post GLP-1 Window with new 2026 human data showing appetite and blood sugar support after stopping the drug.
The supplement industry has spent the last two years trying to beat GLP-1 drugs at their own game. Call your product "Nature's Ozempic", claim it raises GLP-1 levels, and ride the wave. The problem is that strategy is hitting a wall -- legally, commercially, and from a results standpoint. NNB Nutrition is taking a different approach, and it's one the industry should pay attention to.
At a recent industry event, NNB Chief Brand Officer Dustin Elliott and Chief Science Officer Shawn Wells laid out a new category framework they call the Post GLP-1 Window: the period after patients discontinue GLP-1 medications when blood sugar regulation, appetite control, and body composition are suddenly on their own again. The company argues that this is the time for the dietary supplement industry to show its true potential, and their arguments are both sound and scientifically-backed.
NNB's anchor ingredient for this window is GlucoVantage®, their patented dihydroberberine (DHB), and they're now backing the positioning with new 2026 human data. This article explores GlucoVantage from a different perspective: using it after you've cycled off of GLP-1 medications and need to support your body's natural GLP-1 production to keep the benefits coming and the weight off.
Subscribe to PricePlow's Newsletter and Alerts on These Topics
Why "Natural GLP-1 Replacement" Is a Losing Strategy
The regulatory case against GLP-1 replacement claims is already being made in court. A lawsuit against Lemme GLP-1 last year[1] is the cautionary example NNB's team cites directly: go too far with drug-adjacent language and you're defending a claim in court instead of selling product.
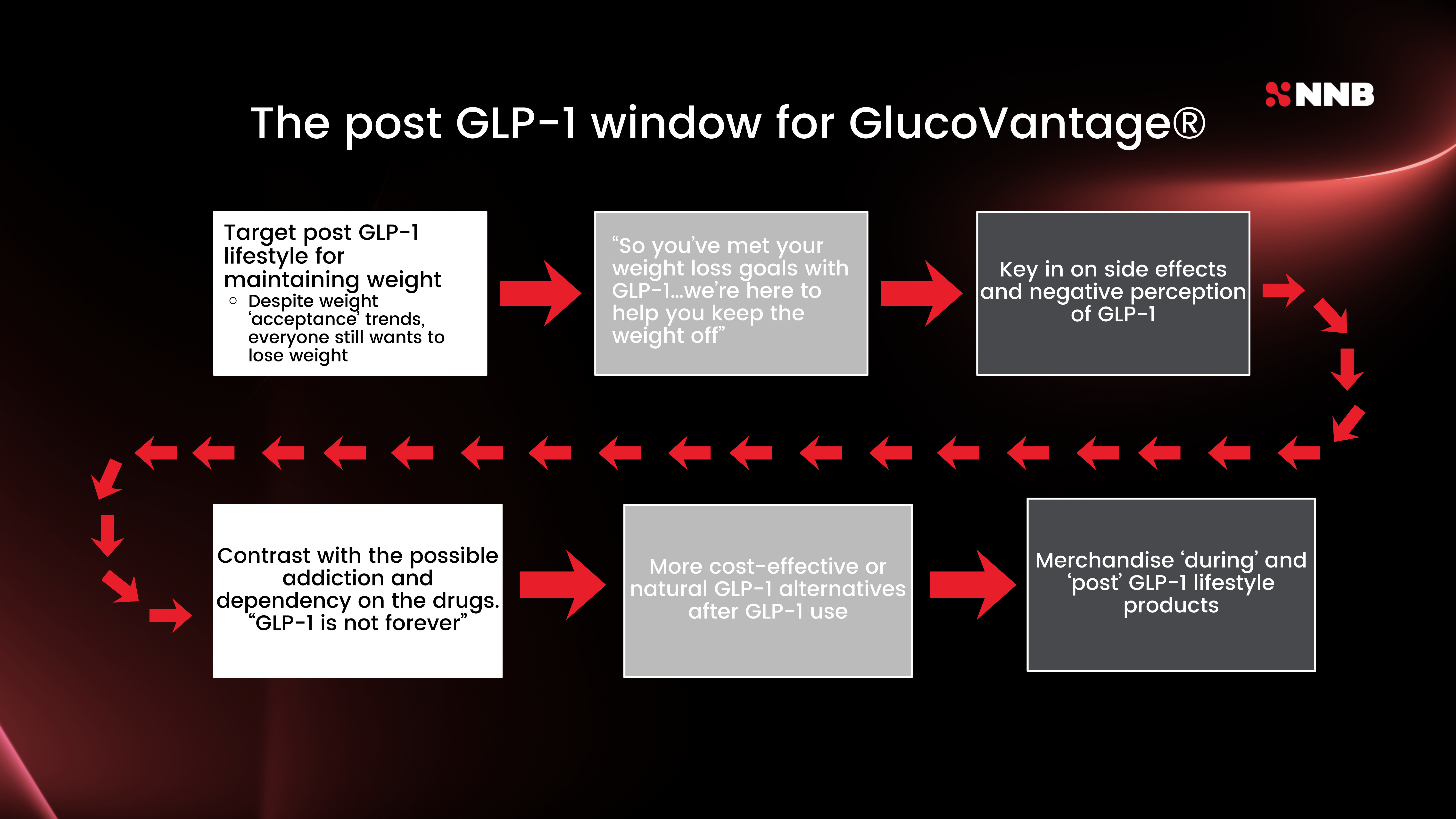
NNB Nutrition's six-step marketing strategy guides brands from targeting post-GLP-1 lifestyle goals all the way through merchandising natural alternatives for users coming off the drugs.
Beyond legal risk, there's the efficacy ceiling. No supplement realistically reproduces what semaglutide, tirzepatide, retatrutide, and other related compounds do at the pharmacological level. Pharmaceutical GLP-1 agonists work through powerful receptor activation that natural AMPK-pathway ingredients can't replicate at comparable magnitude. Brands that promise otherwise set up consumers for disappointment while inviting regulatory scrutiny at the same time.
Then there's what happened to the "GLP-1 support" retail sets. For a period, both GNC and The Vitamin Shoppe dedicated shelf space to GLP-1 support products. The problem was that those sets mostly told consumers to buy protein and digestive health supplements. Consumers learned the lesson and then stopped needing the curated set. The dedicated GLP-1 section has largely collapsed at major retailers because it educated customers right out of the category, and it's easy to find protein, creatine, and HMB in the store.
The Time Window Everyone is Forgetting
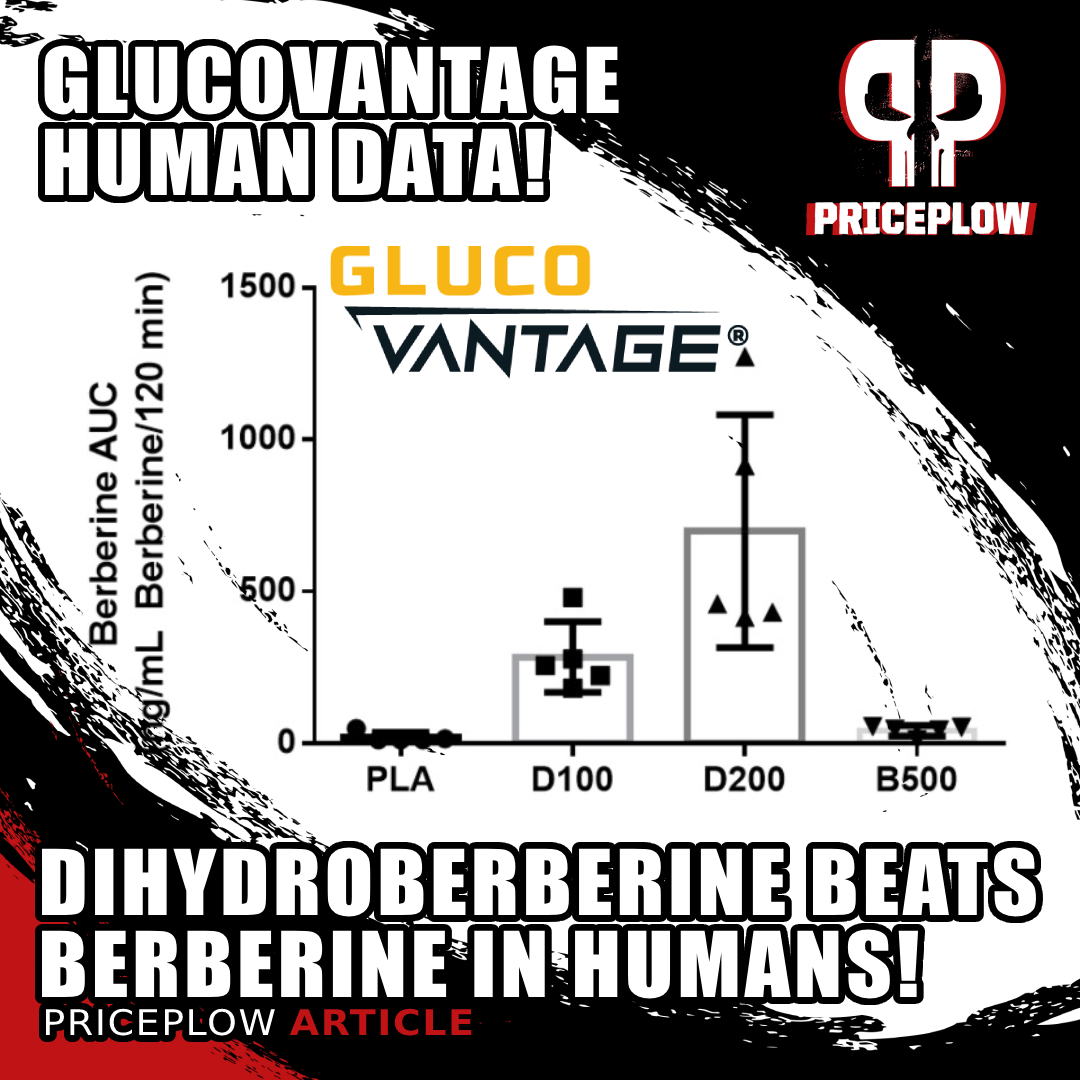
A study published in early 2022 showed that NNB Nutrition's GlucoVantage dihydroberberine outperformed higher doses of normal berberine in elevating plasma berberine and reducing blood sugar and insulin levels!
Here's the market reality NNB is building around. According to FMCG Gurus survey data across the U.S., EU, and Asia, 6% of the U.S. population is currently on a GLP-1 drug. More importantly, 76% of those users are on the medication for less than a year. The drugs are expensive, side effects are real, and many doctors aren't managing titration carefully. Most patients stop cold turkey when they should have considered microdosing in the first place.
Shawn Wells makes the point directly: endocrinologists prescribing GLP-1 drugs today are often less sophisticated about peptide protocols than the biohacker community that's been doing peptide therapy for years. Standard dosing is too high and nobody is slowly ramping down. One week you're at your maintenance dose, the next week you're not, and your body has to figure out the rest on its own.
What follows that cold-turkey stop is predictable: appetite surges back, blood sugar regulation gets more difficult, and the weight that was lost can return quickly (often with less muscle to boot). That transition period is the Post GLP-1 Window, and until now, almost nothing is positioned specifically to address it.
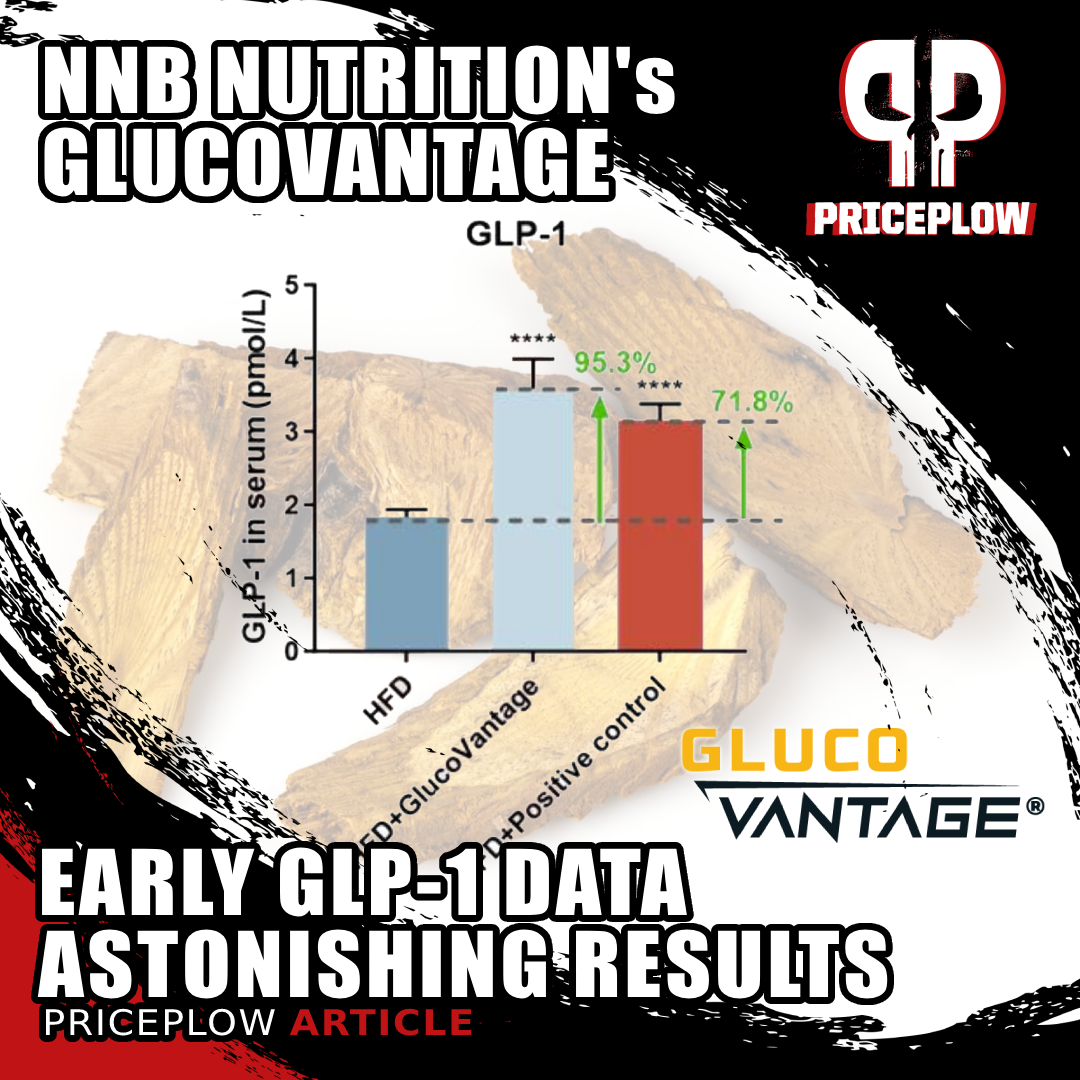
GlucoVantage® dihydroberberine redefines metabolic health by boosting GLP-1 levels by 95% in new research. Its superior bioavailability and multi-faceted benefits—like better body composition, lipid profiles, and oxidative stress reduction—position it as a groundbreaking natural alternative to GLP-1 drugs.
Additionally, the FMCG Gurus data shows that 60% of consumers would pay $50 or more for a supplement delivering 30%+ effectiveness in this space, and 27% would go up to $100. NNB pitched this framing to retailers and the response was positive, not just because the science holds up, but because it's a category regulators can't easily challenge. You're not claiming to replace the drug (which puts you too close to drug and disease claims). You're supporting metabolic function after someone stops using it.
"Post Cycle Therapy" for GLP-1
Think of it as "post-cycle therapy for GLP-1 drugs". The concept will be familiar to anyone who's followed anabolic cycles: when a drug does the hormonal heavy lifting for you, stopping abruptly leaves your body scrambling to re-regulate on its own. GLP-1 "PCT" addresses the same physiological gap, just for blood sugar and appetite rather than testosterone.
Regulatory-safe language for this category includes "post GLP-1 recovery" and "GLP-1 reset support". Both avoid any implication that a supplement replaces or mimics a pharmaceutical, and both have cleared NNB's legal and regulatory review.
Why GlucoVantage Fits This Window
Dihydroberberine does several things, but one mechanism brings the main point here. GlucoVantage doesn't flood your system with synthetic hormones. Berberine ingredient instead supports the body's own GLP-1 production and glucose management through natural pathways, including AMPK activation, PKA pathway signaling, and direct stimulation of GLP-1 secretion from intestinal L-cells.[2-6] When the pharmaceutical's effects are gone, GlucoVantage can support those same pathways at the cellular level.

New 14-week preclinical data on GlucoVantage® dihydroberberine shows 165% GLP-1 increase at week 14 and 33.3% body weight reduction by week 13. Benefits intensified over time rather than plateauing. Natural metabolic support for the post-GLP-1 window.
The bioavailability advantage matters immensely when it comes to these ingredients. Standard berberine has to be converted to dihydroberberine by gut bacteria before absorption,[7] and that's a conversion bottleneck that caps how much actually reaches circulation. GlucoVantage skips that step entirely, arriving already in absorbable form and achieving plasma concentrations roughly 5 times higher than an equivalent berberine dose at just 200mg.[8] It also lasts twice as long (8 hours versus 4) with a dramatically better GI tolerance profile.
For a deep dive into the preclinical data (including the 8-week study showing 95% GLP-1 increases and 27.5% fat reduction versus high-fat diet controls, and the 14-week follow-up where GLP-1 benefits kept building to a 165% peak), see our GlucoVantage GLP-1 preclinical coverage. We won't retread the animal data here. The story this time is human data.
New 2026 Human Data Coming
NNB has two pieces of 2026 human data to report, both of which directly support the Post GLP-1 Window positioning:
-
Acute Data: A Single Dose Reduces Appetite
In a 2026 acute intervention study, participants took a single 200mg dose of GlucoVantage and were assessed against placebo across multiple appetite metrics. The dihydroberberine group outperformed placebo across all five: composite appetite score, desire to eat, hunger, fullness, and prospective food consumption. Subjective appetite decreased meaningfully from a single dose.
A single 200mg dose of dihydroberberine beat placebo across all five appetite measures in NNB's 2026 acute study, including hunger, fullness, and prospective food consumption.
This is notable because one-dose effects are rare with metabolic compounds -- most require weeks of buildup to show a signal. The fact that GlucoVantage moves the needle acutely tells you something about the speed of its mechanism. For someone coming off a GLP-1 drug whose appetite suppression was pharmacologically sustained, a compound that starts working on day one is exactly what the Post GLP-1 Window calls for.
-
42-Day Preliminary Study: Metabolic Divergence
The second dataset comes from an ongoing 42-day placebo-controlled intervention. The full study isn't complete, but the halfway-point trends already show a clear split between groups.
The dihydroberberine group showed what NNB is calling metabolic efficiency: blood glucose stayed stable while insulin secretion decreased, meaning the body was processing glucose more effectively with less insulin required. The HOMA-IR index, a standard measure of insulin resistance, improved in the GlucoVantage group.
At the halfway point of NNB's ongoing 42-day trial, the dihydroberberine group showed stable blood glucose with less insulin needed, while the placebo group trended toward worsening insulin resistance by HOMA-IR measures.
The placebo group showed the opposite, however. Glucose control didn't improve, and both insulin levels and HOMA-IR trended upward, showing movement toward greater insulin resistance rather than away from it.
These are preliminary trends, not final results, and NNB is clear about framing them as such. The complete 42-day data and analysis will give a cleaner picture. But the direction is exactly what you'd want to see for a post-GLP-1 context, where the core challenge is blood sugar starting to drift without the drug's support.
-
Clinical Pipeline
The 2026 acute and 42-day data come alongside two larger randomized trials in progress.
The first is a 12-week, 120-participant, randomized double-blind placebo-controlled trial assessing body composition, glycemic control, energy levels, appetite, and mood in healthy adults.
In an 8-week high-fat diet mouse study, GlucoVantage produced a 9.8% reduction in body weight and a 27.5% drop in fat content, results that tracked closely alongside the positive control group.
The second is a 6-week, 54-participant study with an acute phase and 10 days of continuous glucose monitoring (CGM) data. It measures blood sugar response, insulin response, and GLP-1 response to meals, which are the exact biomarkers most relevant to someone transitioning off a GLP-1 drug.
When both come in, NNB will have a human clinical dossier that credibly backs the Post GLP-1 Window category at retail. That's the infrastructure for a category, not just a single ingredient claim.
The "During" Window: Protecting Muscle
A brief note on something adjacent to the post-GLP-1 story: lean mass. GLP-1 drugs cause significant muscle loss alongside fat loss: one semiglutide study found roughly 70% of total weight lost came from lean body mass, well above what you'd expect from caloric restriction alone.[9] Anyone currently on a GLP-1 drug should be thinking about muscle protection during the weight loss phase, not just blood sugar management after.

Shawn Wells and Dustin Elliott reveal NNB Nutrition's Pure, Potent, Precise revolution at SupplySide Global 2025, discussing precision fermentation, Pürest Creatine, OnSwitch beverage innovation, and the future of pharmaceutical-grade supplement ingredients on Episode #192 of the PricePlow Podcast
DL-185® dileucine is the ingredient NNB has been developing for this kind of application -- triggering muscle protein synthesis even at rest through a mechanism that standard leucine can't replicate. We'll cover the full GLP-1 and lean mass story separately. For now, our DL-185 dileucine overview and the PeptiClear™ concept article are the starting points if you want to dig into dileucine's role alongside GlucoVantage for GLP-1 users.
A Category Built on Real Science
The supplement industry's GLP-1 moment doesn't have to end with enforcement actions, lawsuits, and disappointed customers. NNB's Post GLP-1 Window framework gives brands a category to own that's regulatory-safe, backed by a growing body of human data, and addresses a real physiological need that millions of people will face as they transition off these medications.
GlucoVantage at 200mg twice daily doesn't promise to replace semaglutide. It supports the metabolic function the drug was maintaining through mechanisms the body already uses, and with forthcoming 2026 human data showing it works acutely and directionally over 42 days.
For formulators and brands looking at this space, the groundwork is in place. To stay current on NNB's ongoing clinical results as they come in, subscribe below.
Subscribe to PricePlow's Newsletter and Alerts on These Topics
For more on how GlucoVantage fits into NNB's broader ingredient platform, see NNB Nutrition's Mimetics Platform: When Science Imitates Life and Episode #192 of the PricePlow Podcast with Shawn Wells and Dustin Elliott.





Comments and Discussion (Powered by the PricePlow Forum)