
DMHA: A new workout/focus stimulant that's roughly ~80% the strength of its predecessor, has animal research, and seems to be compliant? Healthy and experienced stimulant users read on!
New year, new stimulant? You bet!
In the wake of the removal of other popular stimulants over the past few years, a battle has raged to fill the void left by the ingredients now gone. Factions of the supplement industry continue to work to replace DMAA, whose fate was ultimately decided in 2019.[1]
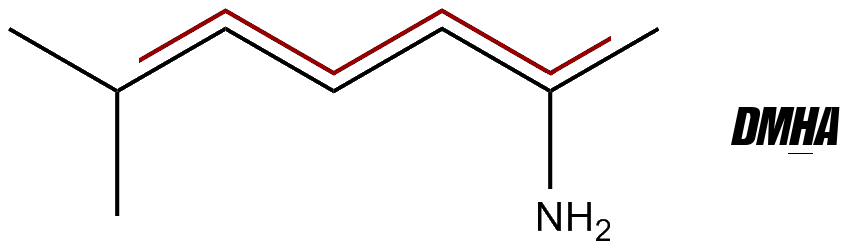
Today we look at the latest stimulant, often labeled as 2-aminoisoheptane but with more specific compound names like 2-amino-6-methylheptane and 2-amino-5-methylheptane. It was slated to be the "next big thing in 2016", a prediction that came true, as the compounds continues to be sold and used through 2023.
Where does DMHA fit into this new era of stimulants, and how does it work? This article dives in, with some legal updates from a 2020 lawsuit against the FDA provided as well.
TL;DR / Summary
-
"DMHA" is actually a set of two different DMAA-like stimulants considered to be ~80% the effects of DMAA.
They work in the same fashion: by boosting dopamine and noradrenaline uptake, while slowing down reuptake long enough for a workout or study session.
- There two versions are named
- 2-amino-6-methylheptane, and
- 2-amino-5-methylheptane
This article primarily focuses on the first one (with a 6, or 'hex'), which has more research. It's called DMHA for short, a term also used by the FDA and legal professionals.
You'll also see it as 2-Aminoisoheptane, but this name that doesn't distinguish between the above two compounds. Other names are Octodrine, 2-amino-6-methylheptane, 6-methylheptan-2-amine, and 1,5-Dimethylhexylamine (hence "DMHA"). See the section on how it works.
-
Potential Side Effects? There's no long-term safety research, although it fared very well in several animals (including mammals), and has an incredibly high oral LD50 in rats.
This class of compounds is less intense than amphetamine, and has far better safety parameters. See the animal research discussing this. It's speculated that it could increase blood pressure, but if that's the case, we predict it would be noticeably less than that of DMAA.
Court filings in 2019 show that there zero related adverse events from 2014-2018.
-
Regulatory: No NDIN (New Dietary Ingredient Notification) has ever been filed on this ingredient, nor are there any known GRAS (generally recognized as safe) affirmations.
However, under DSHEA's definition of what defines a potential dietary supplement, DMHA is referenced as a "constituent of a botanical" since it's found in Kigelia Africana fruit and walnut trees (Juglans regia). See the regulatory section to learn more.
Additionally, in 2019 and 2020, warning letters were sent to twelve companies, with one recipient (Hi-Tech Pharmaceuticals) suing the FDA, who then got the case dismissed with a judge agreeing that an "FDA Warning Letter is not a final agency action" and has no legal teeth. See the Hi-Tech Pharma FDA Lawsuit section for more information.
-
Products Containing DMHA: PricePlow has admittedly not kept up on DMHA-based products. Since Hi-Tech Pharmaceuticals is the company fighting for the ingredient, they're the ones with the most experience to ask.
-
DMHA Suppliers: Manufacturers and brands in countries without compliance issues should contact SYNMR Biotechnology, who claims an 80% Juglans regia extract.
What is 2-aminoisoheptane / 2-amino-6?
- 2-amino-6-methylheptane
- 2-amino-5-methylheptane
This article primarily focuses on the '6' version, or DMHA, due to it being the one that first hit the market and has more research behind it.
It's best-known as DMHA, which is short for one of its other names, 1,5-Dimethylhexylamine. Another name for the 2-amino-6 version is octodrine, which is where much of the research comes from. However, if you see it labeled as 2-aminoisoheptane, it's not 100% clear what exact compound you're getting.
This class of stimulants was first sold in a supplement in 2013,[2] likely with the 2-amino-5 version, but it was quickly removed from the market. In late 2015 and early 2016, however, the similar 2-amino-6 re-appeared in some pre workout supplements and fat burners, quickly grabbing the attention of enthusiasts in the sports nutrition industry as the latest workout stimulant.
As opposed to previous stimulants, DMHA has a greater basis in nature, yet anecdotally still provides the feel, focus, and performance-enhancing effect enjoyed by so many athletes, students, gamers, and even computer programmers.
How it Works: The effect of 2-Aminoisoheptane / DMHA
DMHA shares similar structures to that of DMAA, DMBA (AMP Citrate), and tuaminoheptane (2-aminoheptane), all of which are considered sympathomimetic stimulants or psychostimulants (drugs and/or supplements that promote wakefulness).[3]
Because of the similarities, we can hypothesize how DMHA may exert its effects, however, until more human research is conducted on the compound, emphasis is placed on hypothetical -- yet backed up with anecdotal feedback and animal studies discussed below.
Disclaimer: For healthy, experienced users only!
Immediately we must note that this stimulant is for advanced and experienced users who know their stimulant tolerance well. If you need tons of proven long-term human safety research before using a new ingredient, this is not for you.
It is also not for anyone who does not have a clean bill of health or is on any prescription drugs, and a physician's approval is always recommended when beginning any new diet or supplement regimen.
Interestingly, however, the FDA's warning letters and court cases do not alleged any safety concerns -- this is discussed in the regulatory section below.
The first point of interest is that 2-aminoisoheptane's structural relations are known as monoamine releasing agents, which are compounds that have the capability of increasing levels of certain monoamines.[4,5] These monoamines, most notably dopamine and norepinephrine, are responsible for exerting many of the common effects that stimulants produce – including enhanced euphoria and wakefulness (alertness), respectively.[6]
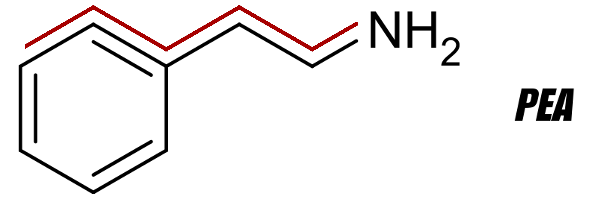
PEA (β-Phenylethylamine) 2D Structure Receptor with PEA Backbone Highlighted. Compare this to the two images below.
In order for a compound to act as a releasing agent, the compounds must be structurally and metabolically related to the classical monoamine neurotransmitters[4,5] (like PEA, pictured to the right).
In the case of DMAA and its structural relatives like DMHA, they share this structure and are able to release both dopamine and norepinephrine through activation of trace amine associated receptor 1, while temporarily preventing their reuptake into the presynaptic nerve. This stops their ability to bind to target receptors, keeping dopamine and norepinephrine active longer, thereby producing the stimulant-like effect that we all know and love.[4,5,7]
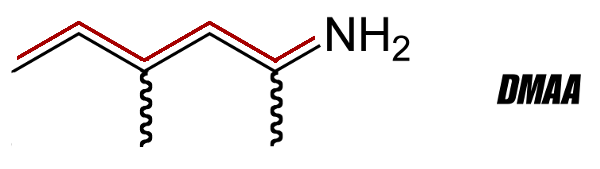
DMAA 2D Structure with PEA Backbone Highlighted
The second common denominator between these three stimulants (DMAA, DMHA, and AMP Citrate) is the addition of a methyl group on the alpha carbon. Pictured above, PEA (β-Phenylethylamine) does not have such components, and is very quickly metabolized, making it less useful for workout purposes. This methyl group addition has three primary effects which are responsible for how the compound will behave within the body. They are:
-
The methyl group acts to limit the effect of monoamine oxidase (MAO), which are a family of enzymes that break down monoamines. By interfering with MAO's ability to breakdown the monoamines, the half life of DMHA should be of decent length, however we have no specific evidence pertaining to this compound.[8]
But be careful of this statement. As alluded to above, we don't want to block MAO for too long, otherwise things start to get real hairy real fast. Case in point: when you have two methyl groups positioned on an amphetamine molecule, you get a drug we all know by the name of methamphetamine, which has disastrous effects.
When it comes to stimulants, too much is never a good thing.
-
The second benefit of the methyl group is that it increases the amphiphilic nature of the compound -- this is where it has both a hydrophilic end and a hydrophobic end. Translation in plain English: a "water-loving" end and a "water-fearing" end, respectively.
Increasing a compound's lipid solubility dramatically increases its ability to pass through the blood brain barrier where it is able to exert its focus-enhancing and euphoric effects.[9,10]
-
The third benefit of the added methyl group is that it boosts the compound's affinity for the catecholamine transporters, thus decreasing the reuptake of norepinephrine and epinephrine.
The more catecholamines that are free to interact with target transporters, the greater the effect of the stimulant. The greater the reuptake, the less catecholamines that are able to interact with target transporters and thus a decreased stimulant effect.[9,10]
We must urge you that there's a lot more at play here, so don't start looking for random stimulants with methyl groups in them. Most fail for one reason or another, but a handful of them exert the right amount of effects for the right amount of time.
Initially classified as a decongestant
Further similarities: Drug suppliers categorize DMHA (labeled as octodrine) as a decongestant.[11] This is exactly how DMAA and ephedrine both started out as well, and also in roughly the same historical timeframe.

Image courtesy the Smithsonian Institute.[12] The instructions from the other images state, "For temporary relief of coughs due to colds. Remove caps. Puncture tube at both ends. Close lips firmly over rounded end, inhale gently twice. Then take 4 to 6 deep breaths through tube, holding each breath several seconds. (If cough persists, discontinue use and consult your physician.)" This was a whopping 350mg dose. Is that jealousy we're feeling here???
In fact, when searching for 2-amino-6-methylheptane, we found that the compound was used as the main active ingredient in Eskays Oralator, which was an inhalant designed to help provide relief for coughs and colds back in the 1950s. The Smithsonian states that each dose was 350mg[12] - more than we recommend in a 24 hour period!
We're not sure what happened to that drug, but it seems to have lost favor due to the rise of ephedrine at the time. Although ephedrine is still legal, it was formally banned in dietary supplements (a long story you can read in our lengthy ephedra). Ephedrine's ban effectively put the industry into a two-decade search for similar stimulants, of which DMHA is the latest.
Now let's turn to what is known from the past animal studies:
Animal Research from the 1940s and 1950s
In 1952, a study published in the British Journal of Pharmacology briefly states the following:[13]
Charlier (1951) showed that the respirations of dogs under chloralose were stimulated by 2-amino-6-methylheptane and that the stimulation lasted for more than sixty minutes.[13]
The chloralose mentioned above is used as a sedative and anesthetic in animals, and DMHA stimulated them for a solid duration of time.
The 1952 study cited above was published about octylamines, a related group of compounds. One of the compounds it focuses on is 2-aminoheptane, or tuaminoheptane, which we mentioned in the beginning of the "how it works" section above since it's a close relative to DMHA (2-aminoisoheptane) here.
In the study, the authors concluded that this related group of compounds had a similar mechanism of action (MOA) of amphetamine, yet less stimulant and cardiovascular activity. Those compounds had nearly as long of a duration time, while being less toxic and having "better" (higher) LD50s.[13]
This type of compound is almost exactly what more aggressive pre-workout formulators are looking for. Note, however, that the scientists were primarily discussing the related octylamines, not DMHA itself. We're getting close, though.
Digging Deeper: Finding DMHA’s Pharmacological Research
Looking back at the 1951 Charlier study that they reference,[14] which was in the Archives internationales de pharmacodynamie et de thérapie (Arch Int Pharmacodyn Ther), we currently run into a dead end and cannot find this paper. We'd love to find it since it's titled "Pharmacology of 2-amino-6-methyl-heptane" - nearly exactly what we want to see.
What's really special about DMHA is that there is absolute, virtually irrefutable data to show it is found in nature in something that has a long, pre-1994 history of being eaten as a foodstuff. -- Bruce Kneller, Giant Sports
The good news is that we fare better with a different study of that same name ("The pharmacology of 2-amino-6-methylheptane") that was published in 1947 in The Journal of pharmacology and experimental therapeutics. PubMed has no abstract,[15] but the journal itself does,[16] and from the full text,[17] we can learn the following:[17]
-
DMHA increased the pain threshold in cats. It has local analgesic properties - and this also worked as an anesthetic in rabbit eyes!
-
When given to dogs intravenously, "2-amino-6-methylheptane hydrochloride exhibited 1/500-1/1000 the pressor activity of epinephrine" -- pressor activity means the ability to raise blood pressure.
Note that epinephrine is a potent pressor (for instance, in humans, just 50ng per kg per minute administration to healthy men raises systolic blood pressure by 17),[18] so more investigation is needed here.
-
2-amino-6-methylheptane HCl does increase cardiac rate
-
Intravenous use in dogs had no detectable effect on the small intestine, detrusor of the urinary bladder, urine secretion, nor respiration.
-
Intravenous LD50 toxicities are listed below:[17]
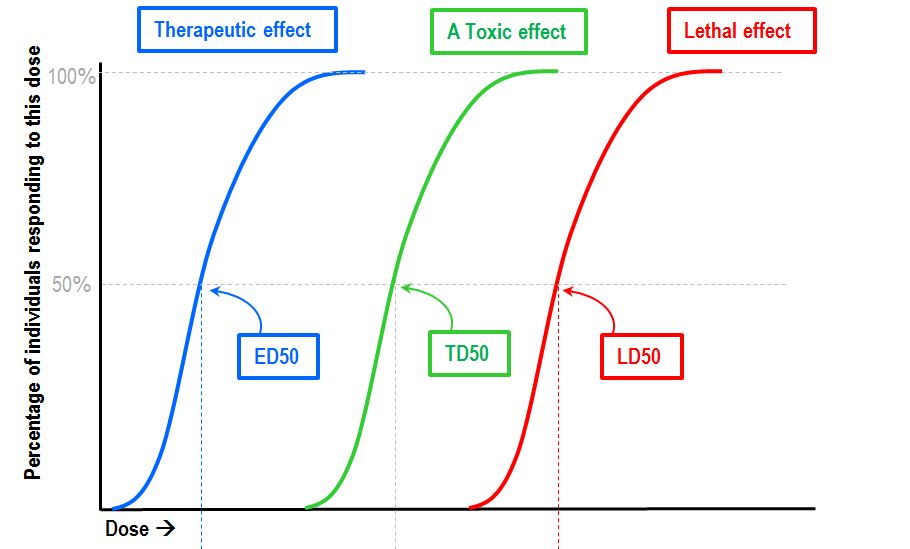
Understand that the LD50 dose is the dose where half of the subjects die. So if you ever take a human LD50 dose of anything, you are literally flipping a coin with your life and need to contact poison control ASAP. We all respond differently, so we stay extremely far away from any such doses.
- Mice: 59mg/kg
- Rats: 41.5mg/kg
- Rabbits: 44mg/kg
- Guinea pigs: 39mg/kg
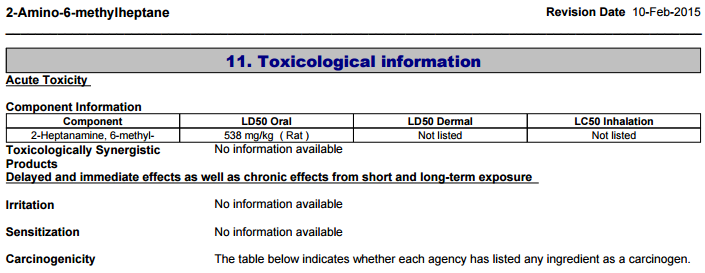
Remember, that's for intravenous use. Oral LD50s are far, far higher (538mg/kg in rats).[19] Also note that their sample sizes were relatively low and they had to interpolate some of this data, so it's not perfect.
-
Autopsies after non-lethal 30-day large doses
"Large" doses were given for 30 days to rats, rabbits, and guinea pigs. The researchers then removed tissue and found that they could not find any demonstrable damage.
These larger month-long doses went as follows:[17]
- Rats: 20mg/kg injected
- Rats: 75 or 100mg/kg daily oral administrations
- Guinea Pigs: 20 or 25mg/kg injected
- Rabbits: 20mg/kg injected
Those are all seriously high doses - the injections were roughly half of the LD50s - yet tissue damage in heart, lung, liver, kidney, spleen, intestine, stomach, brain, and spinal cord could not be found.
Interestingly, the rats orally ingesting those extremely high doses became depressed. Too much of a stimulant is never a good thing.
But the most telling quote from this animal study is in the motor activity section shown below with accompanying chart:
"While there is an indication of more activity after S-51 [DMHA] than absorbed in normal rats, a typical "amphetamine-like" effect was never noted" -- Edwin Fellows[17] (emphasis ours)
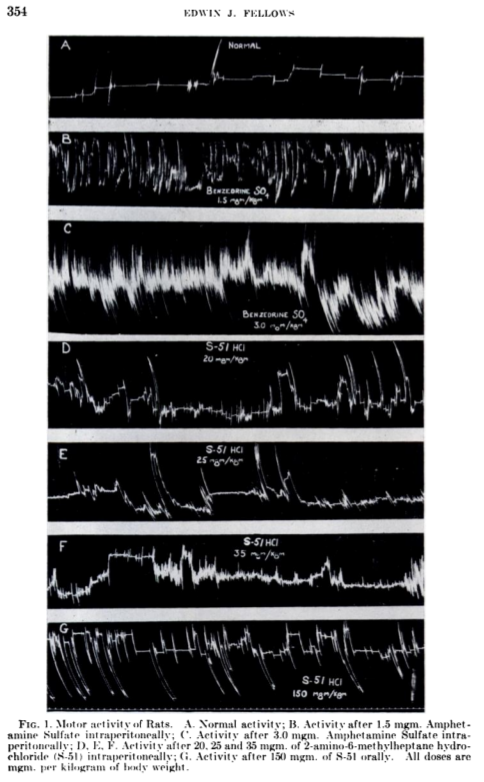
Motor Activity of Rats. A is control. B and C are after amphetamine. D, E, and F are after 2-amino-6-methylheptane administration intraperitoneally (injected into the body). G is after 150mgm orally - an extremely high dose for a mouse. The main point is that DMHA did not affect the rats nearly as much as amphetamine.[17]
In the charts to the right, the researchers looked at the movements across the cage that rats displayed. Their mechanical graphing devices displayed a vertical line anytime they moved across the cage (remember, this was in 1947).
In the control situation, labeled A, the rats are pretty chill, occasionally moving about. In B and C, the rats were given amphetamine, and it clearly shows. In D, E, and F, they were fed intravenous amounts of DMHA, nearly to the point of killing them in F, where they were clearly agitated.
Yet group G was a high oral dose, which shows less franticness and more motor reasonable activity.
Basically, the oral DMHA had the rats moving about, but not banging their heads against the wall like they were with amphetamine.
It's extremely difficult to base human central nervous system activity from rat studies though, so take it all with a grain of salt. It's also worth noting that this ingredient is subjectively less intense than DMAA, which was demonstrated to be relatively safe, provided you used it sensibly and have had no prior cardiovascular conditions.[20,21]
With that said, the data shown is positive and promising for workout aficionados. It lasts a moderate amount of time and is less agitative than amphetamine. From every angle, we keep coming back to the same conclusion - these are the effects that some supplement formulators are looking for.
We'll then discuss dosing, and newly-published research that may have some implications for us in the future.
The stimulant factor – will more dopamine improve mental performance?
While it's hard to say for certain with DMHA, if we can compare it to its structural relatives, then it seems likely that it'll be able to "wake you up", but also more. In saying that, realize that the power of wakefulness could be the dividing factor between achieving a particular task or not.
Can they improve memory?
However, dopaminergic compounds also have other effects that stem far beyond wakefulness, most notably the increased development of episodic memory.[22-24] As the name implies, episodic memory has to do with past episodes or events that shape the way we will respond to similar challenges in the future.[25] As we learn more about which approaches work and which don't through experience, we can apply the same logic to solving new challenges in the future, thus saving time and resources.
For those of you starting to think of this as a work/study/programming aid... you're right on our wavelength.
More than just motivation: bringing the follow-through
Dopamine also has ties to increased motivation and subsequent volition, which is the desire to achieve something and the commitment to doing something, respectively.[25,26]
The key difference between the two lies in the terms desire and commitment – for example, a person may desire to become a professional bodybuilder, but if you throw enough obstacles in their way, the desire to achieve that goal may wane and the end goal never achieved. On the other hand, a person with volition will not stop until the end result is achieved.
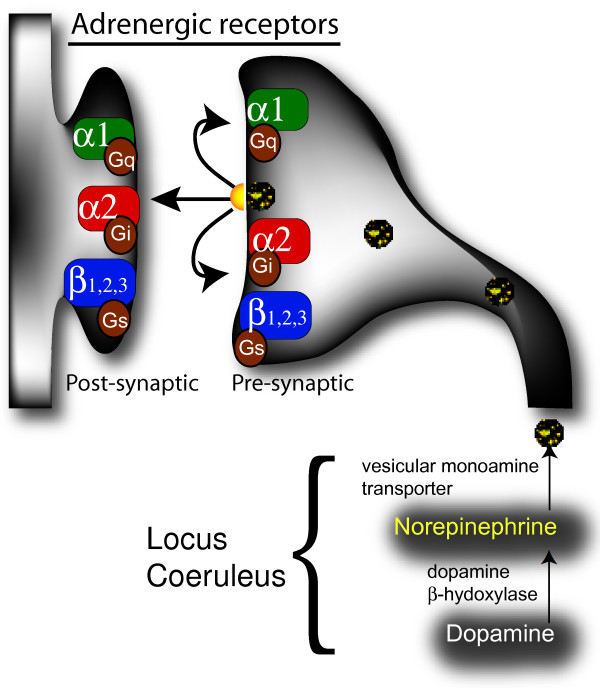
"Norepinephrine is synthesized from dopamine by dopamine β-hydroxylase in neurons of the locus coeruleus. Before the final β-oxidation, norepinephrine is transported into synaptic vesicles by a vesicual monoamine transporter. The vesicles are then transported along the axons comprising the noradrenergic bundle to release sites. At the synapse norepinephrine is released into the synaptic cleft where it binds to various pre- and post-synaptic adrenergic receptors which subsequently activate distinct G protein coupled signal cascades."[25]
It's been stated that there is a motivation 'threshold' where increased motivation eventually turns into volition if a certain level of motivation is reached and surpassed.[26] Those with elevated levels of dopamine work harder for things and remain on task for much longer than those with lower dopamine levels, thus being able to achieve the 'volition threshold'.[24-26]
Lastly, dopamine also plays a key role with the pleasure / reward centers in the brain.[24] In the simplest sense, activation of this pathway tells the person to repeat that action in order to reward again.[23,26] Moreover, activation of this component enhances our memory so that we can remember exactly what we did to achieve that feeling of reward and satisfaction – thus tying in with the above.[24] So not only do we feel better once we achieve something, we remember exactly what we did in order to feel better and are more likely to repeat the task.
But what's the regulatory status?
Regulatory: Basis in nature, FDA warning letters, Hi-Tech fights back, and more
As we've seen in the past, the FDA clearly doesn't "like" ingredients like this, and its compliance case has been in the public sphere for years. DMHA has become part of the long battle over interpretations of DSHEA 1994, the prevailing law of the land in the dietary supplement industry.
First, it's important to note that "2-aminoisoheptane" is not specific to the exact compound. In the past research studies and products found, it had other names, most notably 2-amino-6-methylheptane or Octodrine. Having multiple names is nothing new - after all, there's over ten ways to describe H2O (water).
Second, it's again important to realize that there are two versions of this stimulant, and each have their own compliance case.
DSHEA’s requirements for consideration as a dietary supplement ingredient
In 2016, we were assured that a case does indeed exist for full-blown FDA DSHEA compliance, if some company were to file an NDIN (New Dietary Ingredient Notification) with the FDA.
The default argument for compliance is to state that it's found in nature - as in,
- (A) a vitamin;
- (B) a mineral;
- (C) an herb or other botanical;
- (D) an amino acid;
- (E) a dietary substance for use by man to supplement the diet by increasing the total dietary intake; or
- (F) a concentrate, metabolite, constituent, extract or combination of any ingredient described in clause (A), (B), (C), (D), (E) or (F).[27,28]
Items (C) and (F) are where DMHA would find its compliance factor, as it is said to be a constituent of an herb/botanical. Supposedly, it would need to be further argued that water soluble/hydrolyzable salts of compounds found in an herb or botanical are legal, as the added HCl (hydrochloride) bond makes it into a salt.
Again, however, if it was not sold before 1994, an NDIN would need to be filed with the FDA, and it's highly unlikely they would acknowledge it without argument.
Mr. Kneller has gone on the record to clarify a bit more, stating the following:
"What's really special about DMHA is that there is absolute, virtually irrefutable data to show it is found in nature in something that has a long, pre-1994 history of being eaten as a foodstuff. I am not talking about the algae or the Aconitum species that was (correctly) found this in.
What was not known at the time when the industry first introduced DMHA into the supplement market was that this dietary ingredient has been shown to be found in a fruit-food source.
That is exceedingly good news for enthusiasts... at a minimum, DMHA has a chance of being considered a legit, "meets-the-definition" of the dietary ingredient by FDA.
-- Bruce Kneller
This put us on the lookout for a fruit containing this compound -- that fruit happened to be Kigelia Africana, but it's not the only one out there:
-
Basis in nature: 2-amino-6-methylheptane (Kigelia Africana)
A great look at the Kigelia Africana fruit, which has some very interesting properties... including those from 2-amino-6-methylheptane! Image courtesy Wikimedia commons
A 2014 study shows that Octodrine has a basis in nature: titled "Metabolomic Profiling of Kigelia africana Extracts with Anti-Cancer Activity by High Resolution Tandem Mass Spectroscopy",[29] shows that this fruit contains the compound, as well as several other interesting compounds. The article was published in a well-respected, peer-reviewed journal that really has no stake in the supplement industry.
The fruit interestingly has antibacterial, anti-cancer, and even pain-relieving properties.[29]
But what about talk of J. Regia and walnuts? This bring us to the other form of DMHA (2-amino-5):
-
Basis in nature: 2-amino-5-methylheptane (Jugans Regia)
There's additionally irrefutable evidence that 2-amino-5-methylheptane is found in nature. It's found in walnut bark extract -- not the nut, but the actual bark of the Juglans Regia tree.[30] This evidence is found in the study titled Evaluation of Antimicrobial Properties of Two Different Extracts of Juglans regia Tree Bark and Search for Their Compounds Using Gas Chromatography-Mass Spectrum, where it's named 2-heptanamine,5-methyl-.[30]
Remember, 2-amino-5 is not the same "DMHA" as 2-amino-6 and isn't the basis of most of the research of this paper, but it's obviously very similar. 2-amino-5 is actually reported to have stronger stimulatory effects.
It's worth noting that there's a patent for these ingredients in liquid form, likely a major reason why it's never seen in shots or RTDs.[31]
Speaking of natural occurrence, there's a nut that may contain it too, though, as alluded above:
-
Aconite? Not so fast...
Earlier supplements created in 2013 labeled DMHA as aconitum kusnezoffii / aconite extract,[2] since it's a natural component of the Aconitum plant family. Chances are, it wasn't extracted from this plant when in supplements. Reason being toxicity dangers - this plant family also carries the names Wolf's Bane, Devil's Helmet, and Queen of All Poisons, all of which allude to its lethal toxicity when ingested without proper preparation.[32]
For this reason, the "aconite extract" regulatory argument is basically dead in the water.
Sources state that aconitum contains DMHA, but it's a toxic plant family, so using this in compliance arguments is a riskier endeavor, whether or not you're extracting it.
We can't see the FDA positively acknowledging something labeled as an extract when that plant is clearly toxic. Even if that were technically compliant, it would be too easy for them to claim adulteration, since there is reasonable risk that a true extract from aconite could be dangerous. You'd need to either have a bulletproof extraction process, a slightly risky product that hasn't been perfectly extracted, or simply a fraudulent label. We're not willing to take any of those gambles and we doubt the FDA would either.
Ultimately, if you see "J. Regia" or "Juglans Regia" extract on a label, it's likely you're actually getting 2-amino-5-methylheptane. Point is, both that and 2-amino-6-methylheptane can be found in nature.
The question is, has it been in the food supply, and is that enough for it to be synthetically created and labeled as a dietary supplement without receiving acknowledgement from the FDA as a new dietary ingredient?
This is what may eventually be fleshed out in courts as the FDA butts heads with Hi-Tech Pharmaceuticals:
Hi-Tech vs. the FDA: Warning Letters Do Not Constitute Final Agency Action
In 2019 and 2020, the FDA sent a total of twelve warning letters to brands and manufacturers selling DMHA.[33,34] One such letter was sent to Hi-Tech Pharmaceuticals,[35] alleging that their supplements such as Ultimate Orange, HydroxyElite, Lipodrene Elite, and Synadrene were considered to be adulterated, since the Agency considers the ingredient to be a "New Dietary Ingredient" that has no New Dietary Ingredient Notification (NDIN).[35]
The warning letter requests a written notification within 15 working days explaining the specific steps the brand would take to correct those violations.[35] The response the FDA promptly received may be surprising to newcomers, but not to anyone who's followed the ongoing tiff between these two organizations:
Hi-Tech Pharmaceuticals Sued the FDA over the DMHA warning letters.[36]
Hi-Tech's lawsuit was filed in the District Court of Washington D.C. and requested declaratory and injunctive relief, seeking to ensure clarification that FDA warning letters do not constitute final agency action and carry zero legal weight.[36,37]

A DC District Court Judge ruled that FDA Warning Letters are not Final Agency Action and do not constitute laws or rulemaking.[38]
Additionally, Hi-Tech argues for DMHA's safety and basis in nature (zeroing in on walnut tree / Juglans regia), and states that the FDA is attempting to circumvent official rulemaking policy written by Congress in DSHEA 1994 through the use of warning letters.[36,37]
FDA doesn’t argue: warning letters are informal and advisory
In the FDA's response, they actually agree that warning letters are "informal and advisory" and "communicate the agency's position on a matter" but "do not commit FDA to taking enforcement action" -- they're used to merely "start a dialogue" between the manufacturer and FDA "about the legality of the subject product".[39,40]
With that, the judge (Hon. Reggie B. Walton) dismissed the case on June 29th, 2020,[41] but only after first publishing a memorandum opinion that concludes that "FDA [W]arning [L]etter[s] do[es] not represent final agency action subject to judicial review."[38] The bracketed language means that not only does this warning letter not represent final agency action, but FDA warning letters in general do not, either.
This court cannot decide on DMHA itself
Much of the rest of the case is regarding jurisdictional issues -- the Court "concludes because the FDA Warning Letter is not a final agency action subject to judicial review, the Court lacks subject matter jurisdiction over the plaintiffs' claims".[38]
This means that it's not this court's job to determine whether or not DMHA is actually a legal dietary ingredient - that case is still "unripe" and it would be premature for this court to adjudicate on the matter. This case was really more about jurisdiction, procedure, and the legal scope of warning letters.
However, a few important things are still learned in the court documents:
-
Additional takeaways from Hi-Tech’s FDA DMHA Lawsuit: Safety
While safety and basis in nature weren't the key points of this case, Hi-Tech's filings argue for them anyway.[36,37] Those arguments weren't for nought -- they opened the door to another interesting admission from the Agency:
FDA themselves agree with the statement that the Agency has not stated that DMHA-containing products present a "significant or unreasonable risk of illness or injury"[39] as outlined in one of DSHEA's adulteration clauses.[42] They're instead arguing that it's a New Dietary Ingredient that they haven't been notified about, but not arguing safety.[39]
This is a big revelation for those learning about the ingredient. Hi-Tech's filings state that from 2014 through 2018 (the years prior to their 2019 filing), there were no adverse events related to DMHA -- and there were millions of DMHA supplements sold during those years.[36,37]
FDA also did not argue against DMHA's presence in nature, which seems factual given the data shown in the above portions of this article.
-
Hi-Tech attacks FDA’s attempts to regulate through warning letters
Hi-Tech also attacks the FDA's attempts to remove ingredients from the market through warning letters, since "the law requires FDA to follow certain lengthy steps before the agency can ban dietary supplements".[4,5] Hi-Tech is forcing FDA to uphold its rulemaking procedures (such as those carried out with ephedrine in the early 2000s), as the burden of proof is on the Agency to determine adulteration.[27,28,42]
Instead, Hi-Tech claims that "By issuing the warning letter regarding DMHA, the FDA has sought to further broaden its authority over dietary supplements in direct contravention of DSHEA."[37]
Hi-Tech's lawyers never disappoint, with additional commentary provided:
"The FDA's expanded, aggressive approach to dietary supplement regulation has turned DSHEA on its head, attempting to shift to dietary supplement companies the burden of proving a dietary supplement ingredient is safe and lawful, rather than what is clearly called for by DSHEA, namely that dietary ingredients are foods which are presumed safe and that the FDA has the burden to demonstrate that they are unsafe and/or unlawful."[37]
"As previously noted, Hi-Tech merely seeks to compel the FDA to comply with its statutory obligations that Congress imposed on it in DSHEA. If the FDA intends to impose an industry-wide ban on DMHA as an ingredient in dietary supplements, it must engage in fact-finding and rulemaking"[37]
Returning volley, FDA retorts that they don't need to engage in rulemaking, but could instead proceed through forfeiture, which affords the manufacturer a full range of procedural due process in a federal court.[40]
The judge in this case does not opine on these statements.[38]
-
Hi-Tech attacks FDA’s attempts at using food additive declarations
Finally, Hi-Tech criticizes the FDA's attempts to pigeonhole dietary supplement ingredients that they don't like as "food additives". Reason being, when it comes to food additives, the burden of proof is on the manufacturer. Per the FDA's arguments in this case, "a food additive generally 'shall . . . be deemed to be unsafe' unless FDA has approved its use by regulation and its use conforms to such regulation",[39] citing 21 USC §348.[43]
Hi-Tech argues that "The FDA cannot circumvent these requirements by simply declaring, without proof, that a dietary ingredient is an unapproved food additive. If the FDA intends to ban a dietary ingredient, it cannot ignore its statutory obligations merely because it is required to take a series of lengthy scientific and legal steps."[37]
They later call this food additive declaration to be "arbitrary and capricious and in violation of the requirements of DSHEA, and its actions taken in reliance on that unlawful act are similarly in violation of the law".[37]
The FDA however argues that if DMHA is not a "dietary ingredient" per 21 U.S.C.§ 321(ff) then it may be a "food additive" per 21 U.S.C. § 321(s)(6).[39]
This "adulterated food additive" attack prong has long been used by the FDA in these warning letters, and is something that will likely need to be decided in courts. Hi-Tech is obviously not having it.
Lesson learned: Warning letters are not laws
Ultimately, the point is this: all sides agree that warning letters do not constitute final agency action or rulemaking and have no legal teeth.
Humorously, FDA comments that Hi-Tech didn't even seek retraction, withdrawal, or invalidation of the warning letter.[39] Per the FDA themselves, this "underscores the fact that the Warning Letter carries no legal consequences for anyone, as retraction of the letter would put the parties in no different legal position than they currently hold."[39]
This means that Hi-Tech maintains that DMHA is not "illegal", and if FDA wishes to pursue removal from the market, they will need to do so through their own rulemaking procedures and not through warning letters. Warning letters which, according to the DC District Court, carry no legal weight.[38]
This case is currently dismissed, but without prejudice, meaning Hi-Tech can refile the lawsuit at a later time if necessary. If that were to happen, or there are other DMHA regulatory updates, you can sign up for our DMHA news at the end of this article.
Drug-tested athlete or in the military? Beware, it’s banned by WADA and the DoD
Beyond compliance, we have to talk about banned substances and drug-tested athletes. As of 2016, this was not on the 2016 WADA banned substances list,[44] but it was certainly put there later on, and is on the list as "Octodrine (1,5-dimethylhexylamine)" in the 2023 list published in late 2022.[45]

...don't even think about it!
So drug tested athletes should not be taking this. Even before it was outright banned, WADA has a clause for "similar substances", so technically, DMHA was automatically banned for sport since it is so similar to other banned stimulants.
In addition, DMHA is now on the Department of Defense (DoD) Prohibited Dietary Supplement Ingredients list.[46] As for those with drug-tested jobs, there's a non-zero risk with DMHA, and it's best to ask your drug-test sponsor.
So be smart with this one. Stay away from it if you're a drug-tested athlete or in the military.
Banned in Australia on October 1, 2017
The Australian Government published an advisory stating that DMHA would be banned in Australia on October 1, 2017, as a Schedule 10 substance.[47]
Any side effects?
Consider some of the DMAA safety data
It does seem apt to mention that if it shares a similarity to DMAA, then we can likely expect much of the same effects on heart rate and blood pressure. Note that USPlabs funded a study which examined the effects of 75mg of DMAA alone, 250 milligrams of Caffeine alone and a combination of the two on blood pressure and heart rate.[20]
We urge against proprietary blends with stimulants
When both were combined, which is what we nearly always see in fat burners and pre-workout supplements, they produced small increases in both systolic and diastolic blood pressure.[20,21] This may be problematic for those with pre-existing elevated high blood pressure or whose exercise is likely to cause massive spikes in blood pressure or heart rate, but the numbers were not exceedingly high -- systolic blood pressure was increased ∼6 mmHg with supplement use.[20]
A lot more data was also published in Hi-Tech's DMAA Lawsuit against the FDA -- you can see their DMAA toxicology data, which shows fewer adverse events than vitamin C!
But remember, that is data with a different (yet similar and stronger) ingredient than DMHA -- it's impossible to say more about DMHA until more evidence is published. However, looking at the animal studies and LD50 numbers, it's quite promising:
High LD50 numbers
Note: the following numbers are quite high and should never be even remotely tested.
Fisher Scientific, a laboratory equipment and ingredient supplier, lists 2-Amino-6-Methylheptane as having an oral LD50 of 538mg/kg in rats.[19] Remember, the LD50 is the amount required to cause death in half the animals.
Unfortunately, we don't have an oral LD50 for DMAA to compare to in mice for an apples-to-apples comparison here.
Also, even though this DMHA oral LD50 is quite high, we're not rats. It's a very bad idea to try even a fraction of that amount.
Intravenously - far lower LD50s...
On the other hand, the intravenous LD50s from the original study in 1947 by Edwin J. Fellows were far lower than the oral one cited above - mice have an intravenous LD50 of 59mg/kg.[17]
Compare that to DMAA's LD50 in mice:
The LD50 for methylhexaneamine is 39mg/kg for intravenous and 185mg/kg for intraperitoneal administration (mouse).[48,49]
So DMHA's intravenous mouse LD50 of 59mg/kg[17] deems it to be safer than DMAA in this context -- it takes less DMAA than DMHA to kill half of the mice via IV.
Common sense dictates that you should never be injecting this or any related compound!
Proprietary blends with stimulants are bad for consumers
Our biggest issue is when supplement manufacturers put any stimulants into a proprietary blend and don't allow us to disclose them. Hi-Tech Pharmaceuticals has always been obliging of providing doses when asked.
If we can't know our ideal doses, and the various products all have differing doses, it ultimately puts the consumer in a more speculative situation than necessary. We always encourage supplement brands to fully disclose the stimulant amounts on their labels, and at the very least, allow us to disclose them online on PricePlow for our like-minded readers.
Court documents: No adverse events from 2014 to 2018
Finally, court documents from Hi-Tech Pharmaceuticals' lawsuit against the FDA concerning their warning letters (discussed below in the Hi-Tech Pharma FDA Lawsuit section) show that there were no adverse events from DMHA-containing supplements from 2014-2018 (the documents were filed in 2019),[36,37] and those years had heavy use of the ingredient.
All in all, if using one of these products, assessing tolerance by using half of a serving is a smart way to see how it works for you. Wear a heart rate monitor. Take your blood pressure. Be cautious, smart, and get a doctor's approval.
Conclusion: DMHA, J. Regia, and the potential future of “Octodrine”
As interesting as these studies are, they might eventually spell bad news for supplement companies using the 2-amino-6 version.
In 2015, two studies were published showing the following:
-
Octodrine displays a wide variety of anti-microbial and anti-fungal activity.[50] It "was shown to be one of the most effective drugs in killing serum-grown Candida albicans without significantly affecting the survival of host macrophages and skin cells."
-
Octodrine killed a Uropathogenic strain of E. Coli bacteria after five days of exposure, and within a week, it killed everything the scientists tested for.[51] It didn't perform the "best", but it could still have use in treating urinary tract infections (UTIs).
The study from the first bullet point raises some concerns - not safety-wise, but instead in terms of new dietary ingredient applications and marketability. They stated that while octodrine had been used as a decongestant, it was the one compound in the study that'd never been tested to kill fungi. Low and behold, it turned out to be the best one.
Combine this with the fact that there's a patent tied to the first study above[50] titled Method of treating microbial infections,[52] and you can begin to see a concern: there's clearly potential for Octodrine as a prescription antibacterial, antifungal yeast infection drug, and possibly as a UTI treatment too. With that said, if an NDI is submitted before an IND (Investigational New Drug) application, then the dietary supplement case would win out.
This could be a major reason why companies like Hi-Tech Pharma have focused on Juglans Regia based labels, which is presumably the 2-amino-5 version of the ingredient.
While PricePlow is no longer covering news pertaining to DMHA-based products (you can follow Hi-Tech Pharma for those), we will keep the industry aprised on the legal ramifications. To our best knowledge, in three years since the courts ruled that the DMHA warning letters do not have legal teeth,[38] no further action has been taken on this ingredient.
Meanwhile, for international brands/manufacturers without compliance concerns, the most long-standing raw material supplier of this ingredient is SYNMR Biotechnology, who has stated that they have a juglans regia variant with 80% standardization strength.
If there are more legal updates, you can stay up to date by signing up for our DMHA-based news alerts below:









Comments and Discussion (Powered by the PricePlow Forum)