Did you ever buy Premier Protein Shakes between August 8, 2011 and October 12, 2018?
If so, then you're entitled to some money back -- up to $40.00 sent in the mail!
The important links
Cutting to the chase, let's get you to the settlement (and be sure to share this with friends who you know have bought these products):
- Go to the Lawsuit Settlement Claim Page to claim your benefits
- Read the second amended lawsuit (PDF) with lab tests
- Read the full settlement notice with key dates and FAQs
Watch Mike's video explaining the situation
More explanation is below, but if you'd rather watch and listen than read, check out the video and subscribe to our YouTube channel, since another big one's about to drop:
What happened?
In August of 2017, a class-action lawsuit was filed against the Premier Nutrition Corporation, the company that makes not only Premier Protein Shakes and Bars, but also PowerBar, Supreme Protein, and Joint Juice.
The lawsuit (linked above) alleges that the Premier Protein Ready-to-Drink (RTD) Shakes were shown to only contain between 26.9g and 28.34g protein, not the 30g specified on the label.
They provided lab tests to show this -- one such lab test is below:
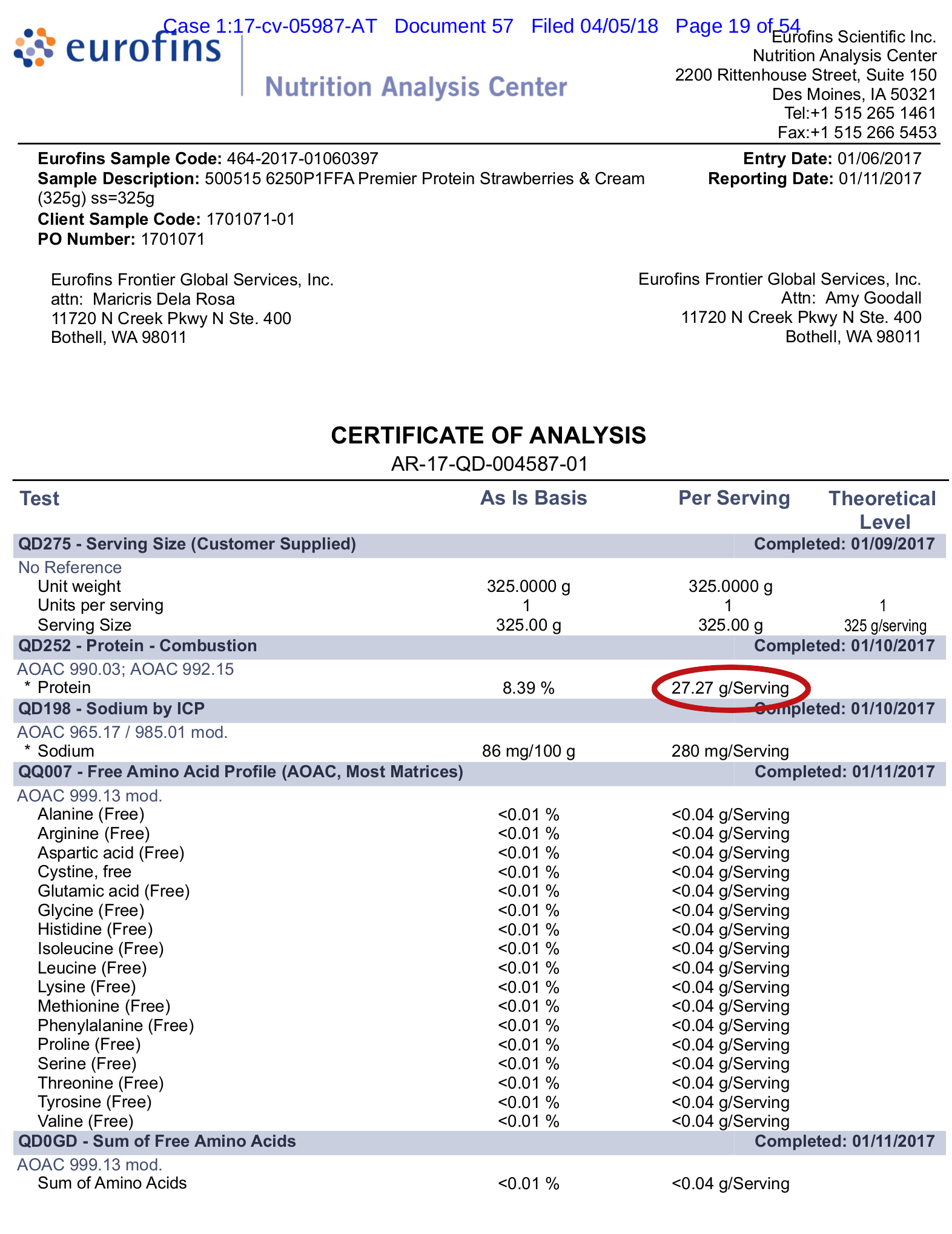
One of several lab tests shown in the legal complaint. This one tested out at 27.27g rather than 30g. Note that there was no amino acid spiking!
After a year of legal proceedings, the Premier Nutrition Corporation decided to settle to the tune of 9 million dollars. But we must add that the Premier Nutrition Corporation admits no wrong-doing despite the large settlement amount.

The Premier Protein Lawsuit has been settled, and that means customers who bought these RTDs can claim up to $40 in benefits!
If the Court approves the Proposed Settlement in early 2019, it will be finalized and customers who have been added to the class will receive their benefits in the mail.
Protein under-dosing -- but not amino acid spiking
Important to note is that this is not an amino acid spiking scam -- as you can see in the lab tests, no free form amino acids were added to the product. It is simply based upon allegations of under-dosed protein.
Is less than 10% under spec illegal though?
You might ask, is 28.34g protein in a 30g bottle really that bad? It's less than 10% under!
Despite our leniency on our YouTube channel, the answer to that question is yes.
Protein powders and protein shakes are not truly "natural" products. This isn't something like milk that can vary -- the protein blend in those Premier Protein blends don't just come out of cows.
Instead, the protein is fortified, and that makes it a Class I Nutrient.
According to the FDA's Guidance for Nutritional Labeling:[1]
Class I nutrients are those added in fortified or fabricated foods. These nutrients are vitamins, minerals, protein, dietary fiber, or potassium. Class I nutrients must be present at 100% or more of the value declared on the label ; in other words, the nutrient content identified by the laboratory analysis must be at least equal to the label value.[1]
-- United States Food and Drug Administration
Before that, the document shows where you can find this in law -- "FDA regulations define two nutrient classes (Class I and Class II) (21 CFR 101.9(g)(3)) and list a third group (Third Group) of nutrients (21 CFR 101.9(g)(5))."
Looking for 21 CFR 101.9(g)(3), we find the following:[2,3]
(i) Class I. Added nutrients in fortified or fabricated foods;
And then at 21 CFR 101.9(g)(4):[2,3]
(i) Class I vitamin, mineral, protein, dietary fiber, or potassium. The nutrient content of the composite is at least equal to the value for that nutrient declared on the label.
Since protein is added / fortified in this food, which is indeed fabricated, it applies here... and it applies to every protein powder you've ever bought, unless they found some magical cows that are lactating pure protein powder!
So now what?
If you believe you deserve Settlement Benefits, you can click the links posted above to get added to the Class and wait. You can also get involved and leave comments or even show up in court if you believe the Proposed Settlement is too big or too small!
Until then, you should definitely sign up for our protein news alerts below, because the Lenny and Larry's Lawsuit Settlement is coming soon. Stay tuned.
Protein – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.





Comments and Discussion (Powered by the PricePlow Forum)