
Most joint supplements measure how your knees feel. Kinexa measured what is actually happening inside. Cartilage, ligaments, menisci, synovium, and subchondral bone. All five. One study. MRI-confirmed.
For the past 25 years, the joint health supplement category has largely asked one question: "How does it feel?" We see comfort scores, pain questionnaires, and subjective stiffness ratings. These are meaningful outcomes, and there's nothing wrong with wanting your knees to hurt less. But they don't tell you whether the structures inside your joint are actually holding up.
Joints aren't just pain sensors. They're load-bearing mechanical systems. Cartilage, menisci, ligaments, synovium, and the bone beneath the cartilage each degrade on their own timeline, often well before you feel a thing. By the time symptoms show up, the underlying tissue damage has usually been accumulating for years.
Kinexa: PLT Health's New Joint Support Ingredient with a Bombshell Study
There is a throughline connecting underlying joint structure with the experiential aspect of how the joint feels and the functional aspect of how the joint performs. This holistic approach to the whole joint is exactly what Kinexa Joint Longevity Complex, a new patented botanical ingredient from PLT Health Solutions, was designed to address. Backed by a groundbreaking six-month randomized, double-blind, placebo-controlled clinical study using three types of MRI imaging, Kinexa is the first dietary supplement ingredient to demonstrate statistically significant improvements across five distinct joint structures simultaneously, alongside improvements in strength, function, and subjective comfort.
Research Unlike Anything We've Seen in Dietary Supplements

PLT Health Solutions: Growth Through Innovation
As PLT's Director of Medical and Scientific Affairs Jeremy Appleton, ND put it: "Comfort is how the joint feels today, but structure is what determines whether it keeps working years from now."
To put it simply, this new research study is unprecedented, and Kinexa is set to take the joint supplement space by storm... and in a hurry.
Before we get into the details, sign up below for PricePlow's PLT Health news alerts so you don't miss what's coming next:
Subscribe to PricePlow's Newsletter and Alerts on These Topics
What Is Kinexa?
Kinexa Joint Longevity Complex is a patented, low-dose formulation of three standardized botanical extracts with deep roots in Ayurvedic medicine:
- Haritaki (Terminalia chebula fruit extract), standardized to chebulagic and chebulinic acids (the "King of Medicines" in traditional Indian herbalism), is known for its antioxidant and cytoprotective properties.[1,2]
- Turmeric (Curcuma longa rhizome extract), standardized to curcuminoids, is widely recognized for supporting healthy inflammatory balance.
- Indian Frankincense (Boswellia serrata oleo-gum-resin extract), standardized to amyrins, is a workhorse anti-inflammatory synergist that PLT has studied extensively across multiple Boswellia ingredient platforms.
The clinical dose is 200mg once daily. The three components are all "Old Dietary Ingredients" (pre-DSHEA, grandfathered in under the 1994 Dietary Supplement Health and Education Act), so the formula carries a favorable regulatory profile for US formulators. Kinexa is certified gluten-free, non-GMO, Kosher, Halal, and vegan, and it's compatible with soft gels, capsules, tablets, stick packs, powders, chews, and effervescents.

Kinexa combines haritaki, turmeric, and boswellia standardized to chebulagic and chebulinic acids, curcuminoids, and amyrins respectively, at a single 200mg daily dose.
Why Most Joint Research Doesn't Tell the Whole Story
If you look at the clinical evidence behind most popular joint health ingredients, there's a pattern. Study populations are asked whether their knees feel better. Some studies add a functional assessment like a walk test. A few look at biomarkers such as urinary C-terminal telopeptide of type II collagen (uCTX-II), a cartilage breakdown product that shows up in urine. But it is rare for studies to include actual MRI of the cartilage, and almost unheard of for imaging to be done on other joint structures, such as subchondral bone, synovial tissues, or ligaments.
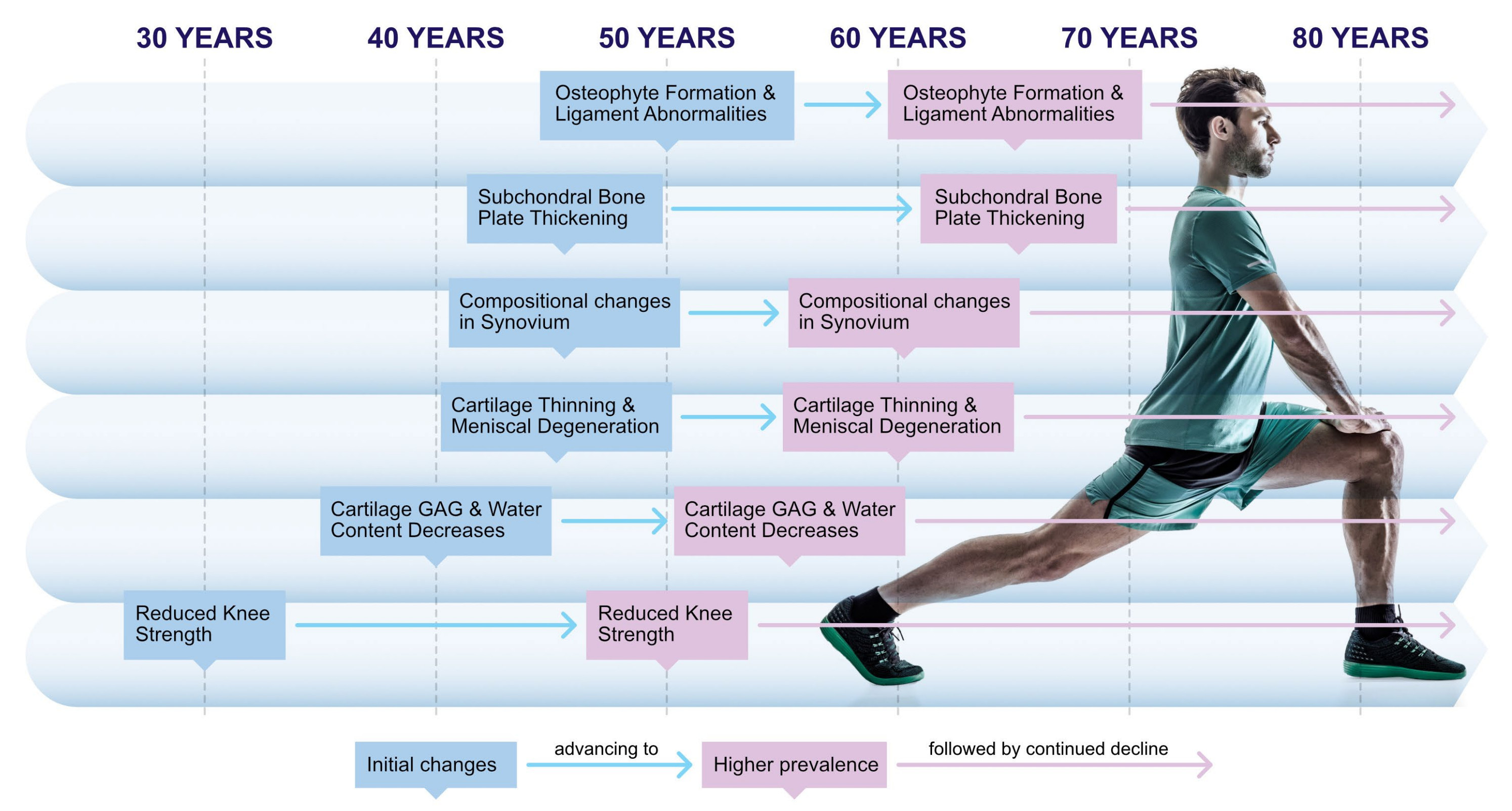
Cartilage GAG loss and reduced knee strength begin as early as the 30s, with osteophyte formation and subchondral changes following through later decades.
But measuring one tissue, or asking people how they feel, doesn't tell you what's happening inside the joint as a whole. For instance, the menisci are taking load, the ligaments are maintaining stability, and the synovium is lubricating the joint and feeding nutrients to the cartilage. The subchondral bone (the dense structural layer directly beneath the cartilage) is absorbing shock and adapting to mechanical load. When the subchondral bone develops abnormalities (such as bone marrow lesions, cysts, osteophytes, and attrition), those changes affect joint stability and may contribute to pain directly, as in the case of osteophytes (bone spurs) which can protrude into surrounding innervated soft tissues..
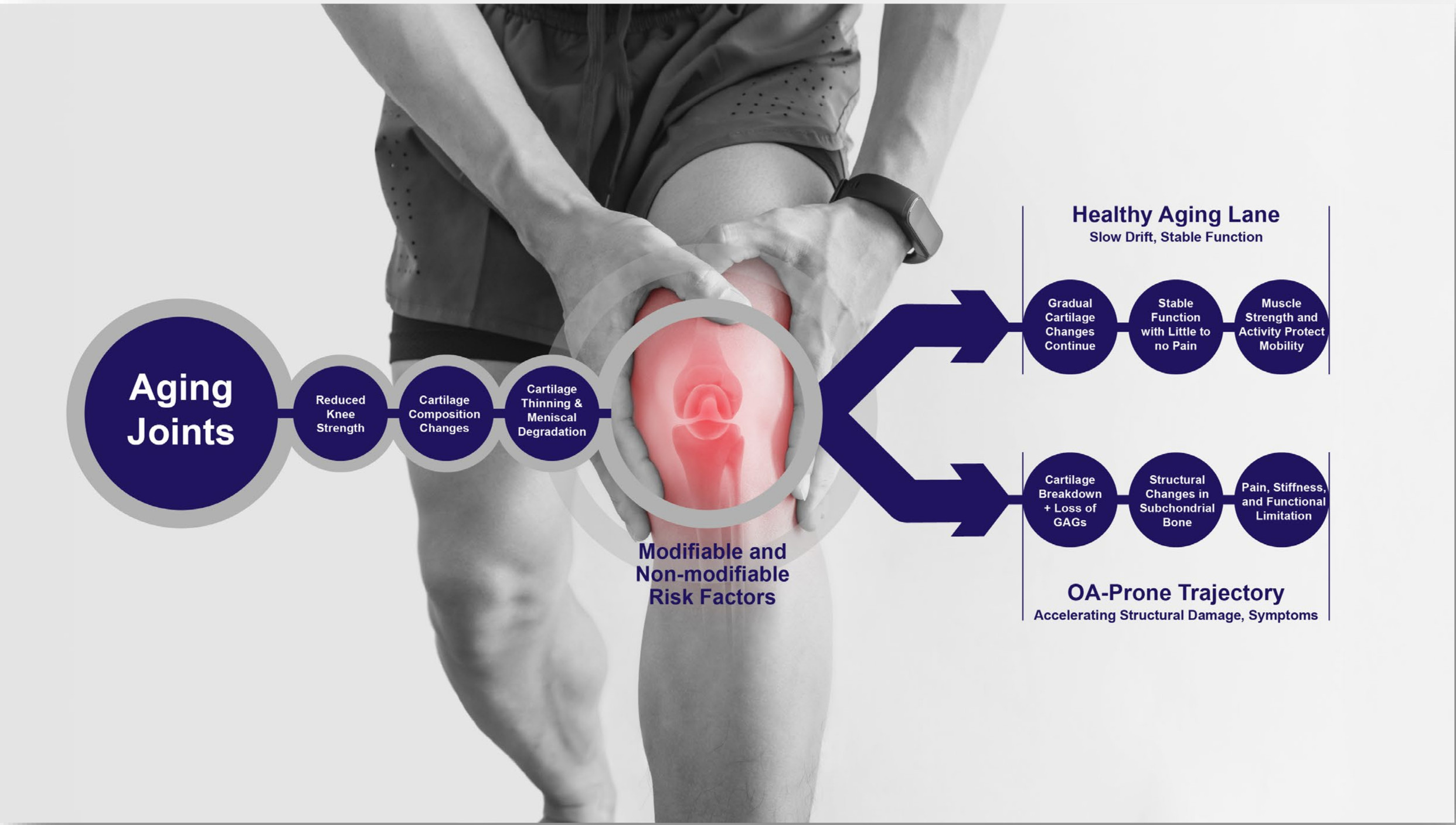
Aging joints split toward stable function or accelerating osteoarthritis depending on modifiable and non-modifiable risk factors acting on cartilage, menisci, and bone.
Evaluating all of these structures together, in living subjects taking a dietary supplement, is extraordinarily rare. It requires expensive, sophisticated imaging protocols that orthopedic specialists sometimes use to assess the health of the joint, but that the supplement industry has almost never pursued. That's what PLT invested in for Kinexa.
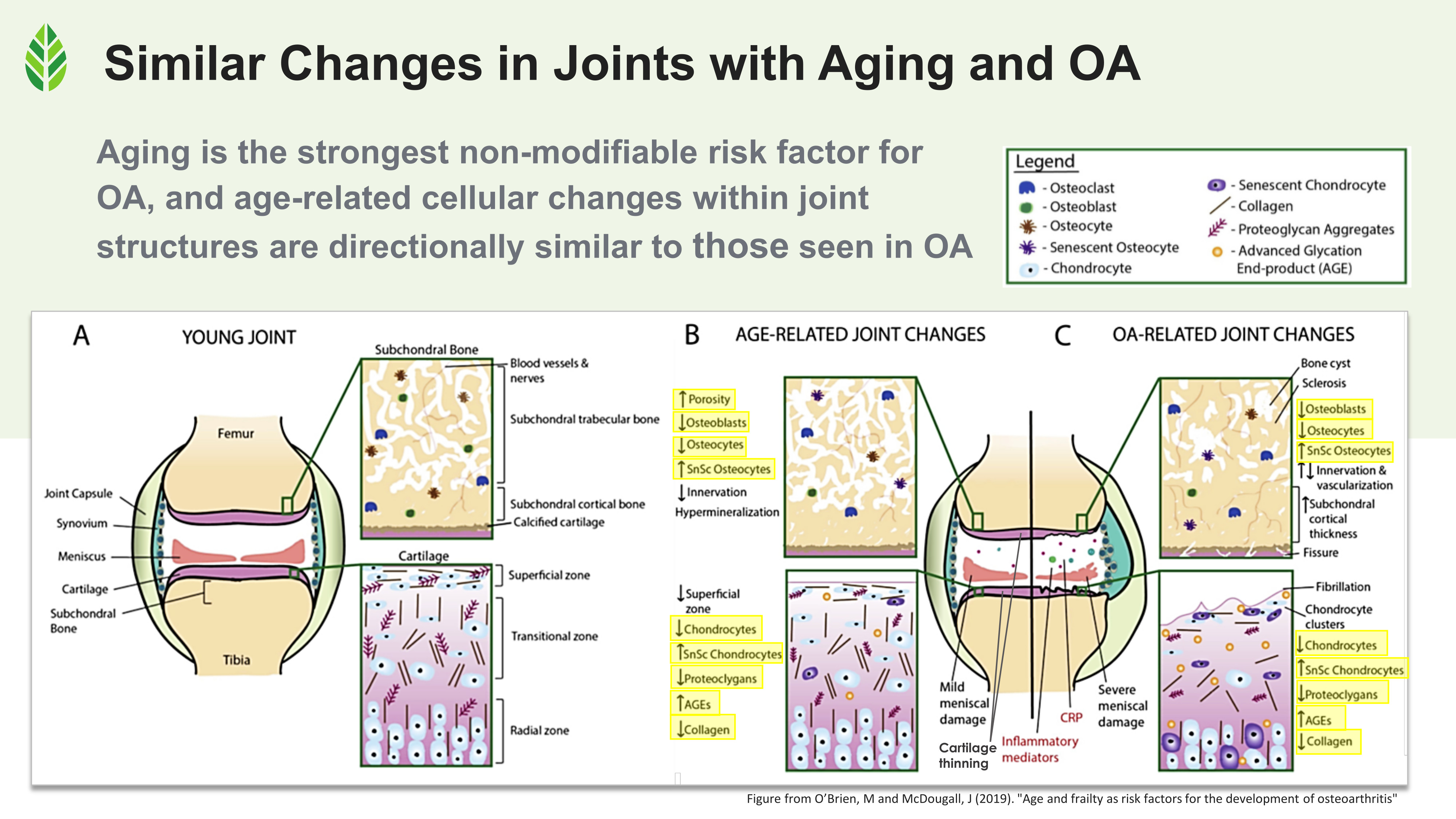
Cellular changes in aging joints and osteoarthritis-affected joints mirror each other across subchondral bone, cartilage, and synovium, making age the strongest non-modifiable osteoarthritis risk factor.
The Clinical Study: An Unprecedented Look at the Whole Joint
Study Design
The Kinexa joint structure study enrolled 100 subjects (50 men, 50 women) aged 40 to 75 years (mean age 50), with a BMI of 20 to 29 kg/m², and a Grade II or III Kellgren-Lawrence classification of knee osteoarthritis (OA). Subjects were randomly assigned to 200mg Kinexa or matching placebo daily for 180 days. Assessments occurred at baseline and at days 30, 60, 120, and 180. Imaging was conducted at baseline and day 180.
The study used four categories of outcome measures:
- Subjective assessments: Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Visual Analog Scale (VAS) for pain, and Lequesne Functional Index (LFI)
- Functional testing: Isometric knee extensor torque, Six-Minute Walk Test (speed and distance), and Timed Up and Go test
- Biomarkers: High-sensitivity CRP (hsCRP), matrix metalloproteinase-3 (MMP-3), and urinary CTX-II (uCTX-II)
- Imaging: Semi-quantitative MRI for cartilage volume and thickness; delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) for glycosaminoglycan (GAG) composition; and Whole-Organ Magnetic Resonance Imaging Score (WORMS) for comprehensive whole-joint structural assessment
What do WORMS and dGEMRIC Measure?
WORMS (Whole-Organ Magnetic Resonance Imaging Score) is a validated, semi-quantitative MRI scoring system that evaluates structural changes across multiple regions of the knee joint, including cartilage morphology, ligament and meniscal integrity, synovial health (synovitis, effusion, cysts, loose bodies), and subchondral bone (bone marrow lesions, cysts, osteophytes, and attrition).[3] It's one of the most comprehensive tools available for characterizing joint structure, and it's rarely used in dietary supplement research because of the cost and complexity involved.
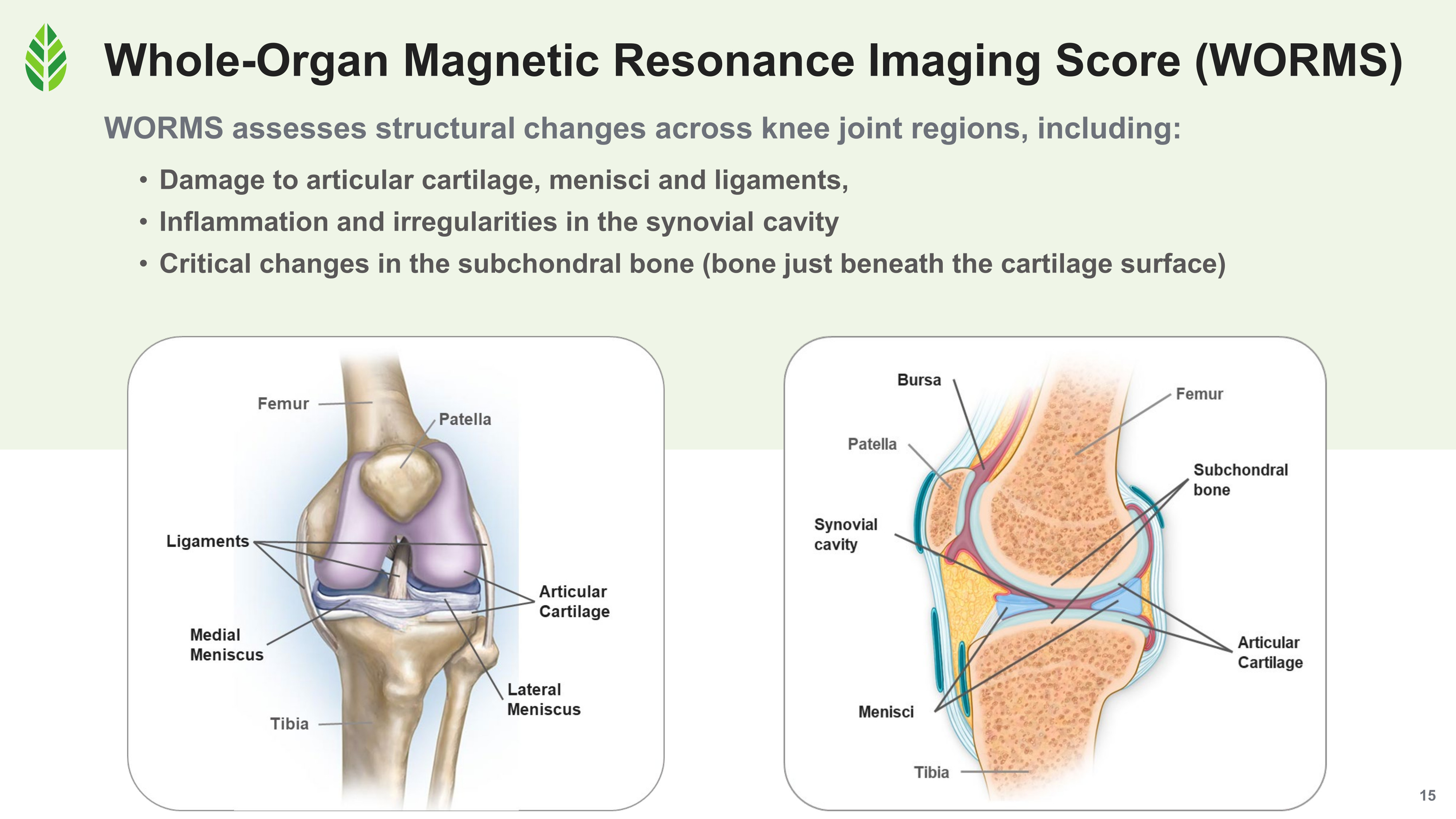
WORMS evaluates cartilage, ligaments, medial and lateral menisci, synovial cavity, and subchondral bone across the whole knee in one validated semi-quantitative scoring system.
dGEMRIC uses a gadolinium contrast agent that penetrates cartilage tissue in inverse proportion to its glycosaminoglycan (GAG) content.[4] Healthy cartilage, with dense GAGs, absorbs less gadolinium and produces a higher index score. Degraded cartilage lets more gadolinium in. This technique gives researchers a direct, quantitative window into cartilage biochemical quality, not just its physical dimensions.
All in all, this was an exhaustive study, the likes we've never seen before in the dietary supplement industry. Now on to the results:
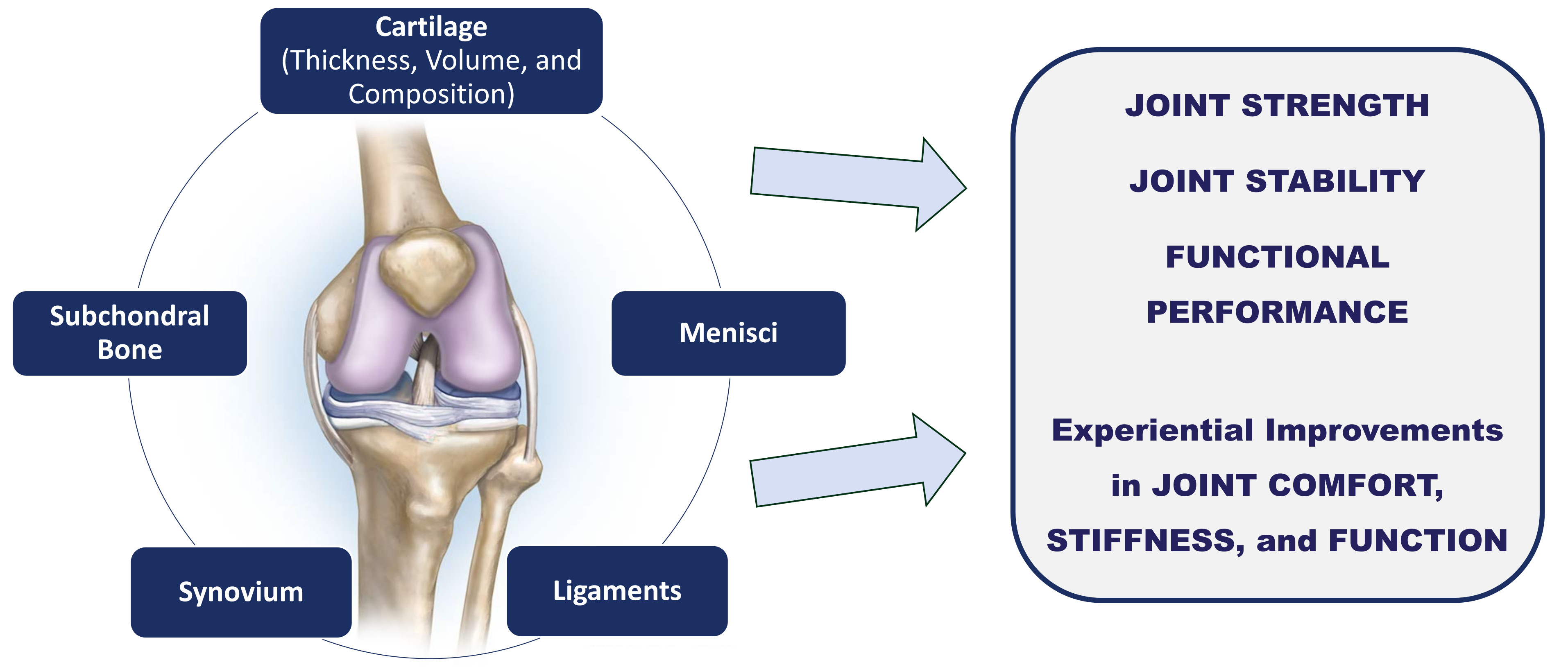
MRI-confirmed improvements across all five joint tissues feed into real-world gains in joint strength, stability, functional performance, and subjective comfort.
Clinical Results: Five Structures, One Supplement
Cartilage: Thickness, Volume, and Composition
After 180 days, cartilage thickness improved 10% in the Kinexa group relative to baseline, and the improvement was statistically significant versus both baseline and placebo. The placebo group, tracking the expected natural progression of Grade II/III OA, showed significant cartilage thickness decline over the same period. Cartilage volume significantly improved versus baseline in the Kinexa group while significantly declining in the placebo group, following the same diverging pattern.
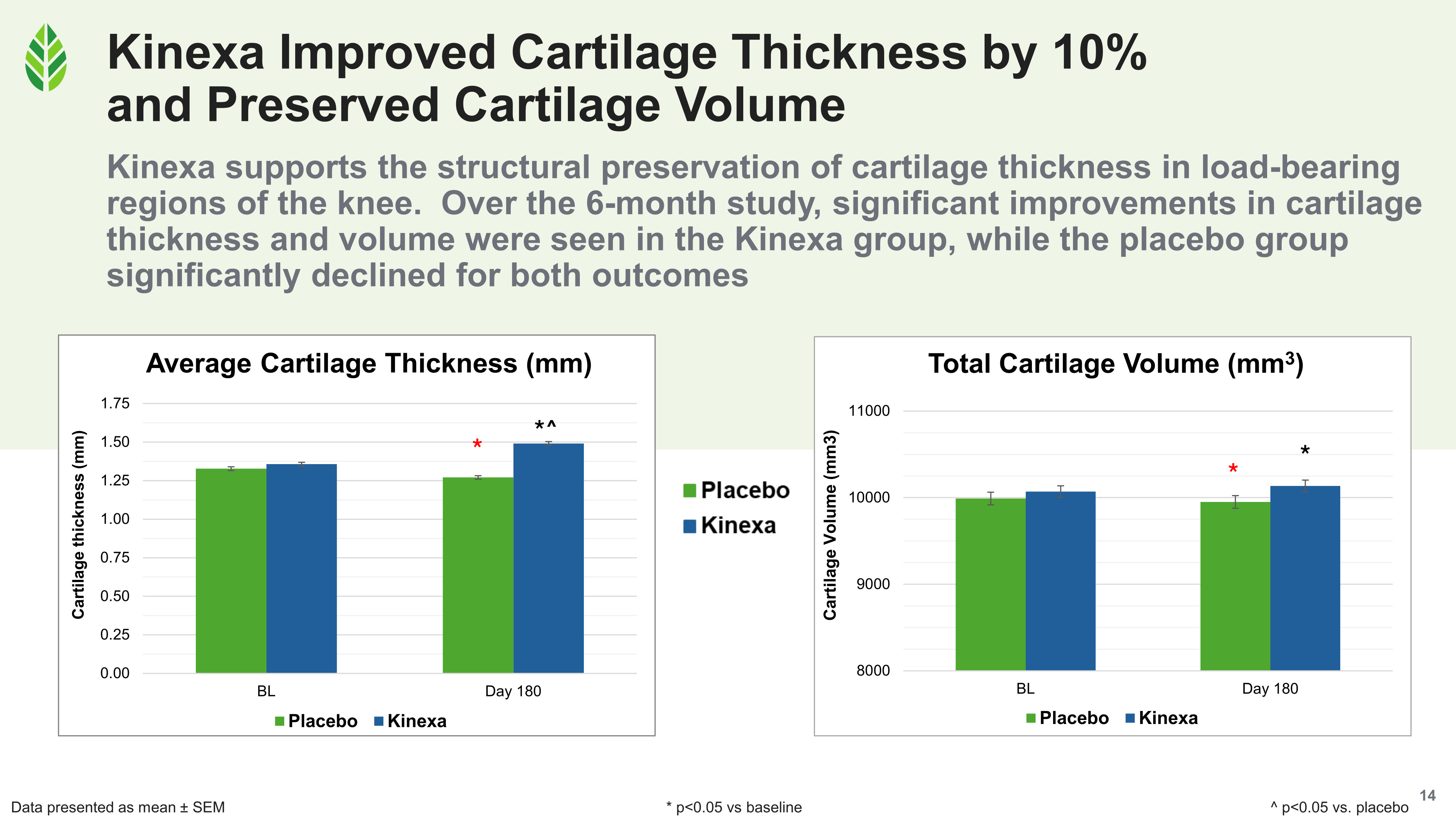
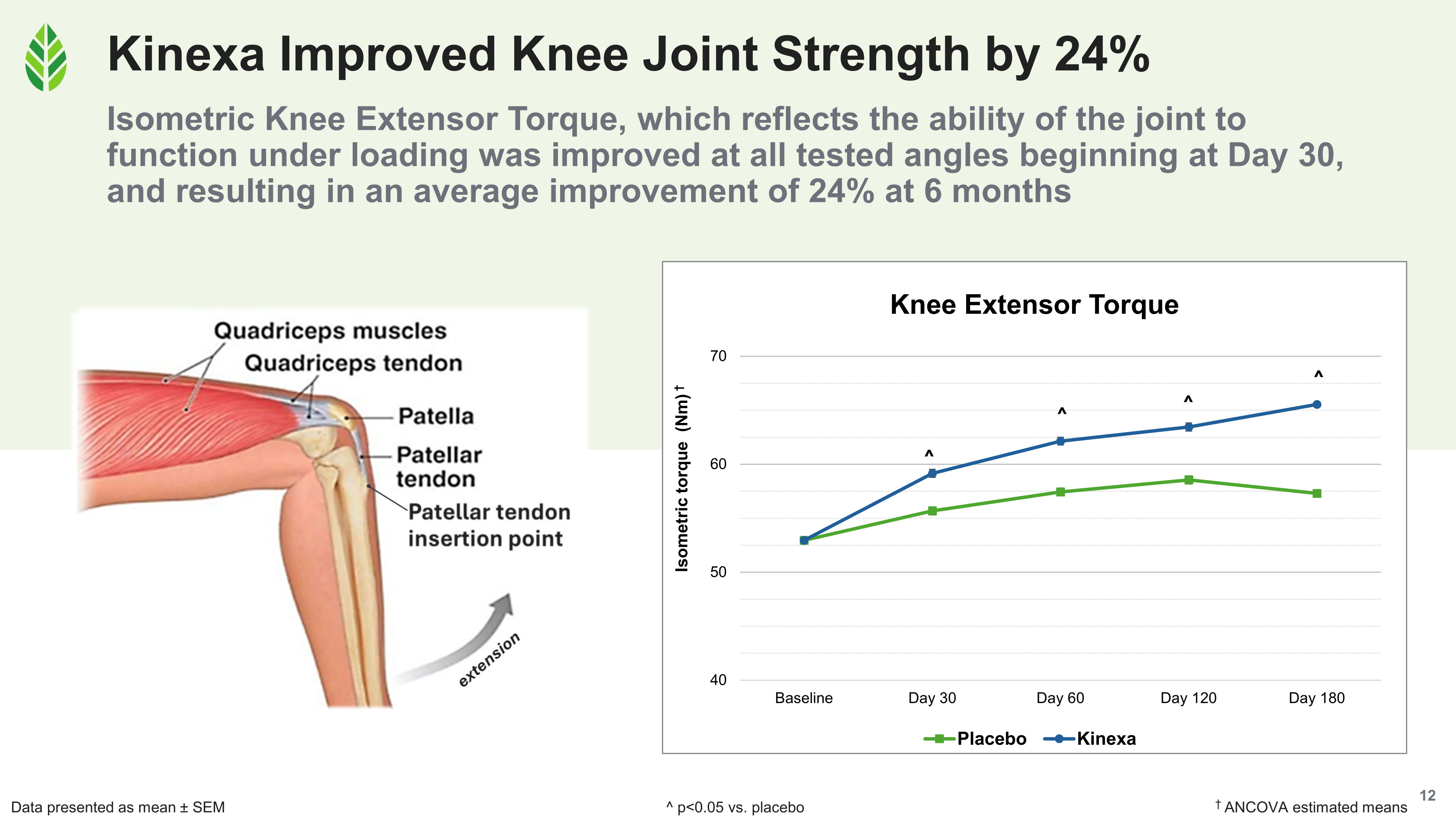
Cartilage thickness grew 10% and volume held in the Kinexa group at day 180, while the placebo group declined significantly on both MRI measures.
Cartilage composition, measured by dGEMRIC, improved 4% in glycosaminoglycan concentration in the Kinexa group (significant vs. baseline and placebo), while the placebo group's GAG levels declined significantly. This matters because GAG content is a direct marker of cartilage functional quality: its water-holding capacity and its ability to resist compressive load.
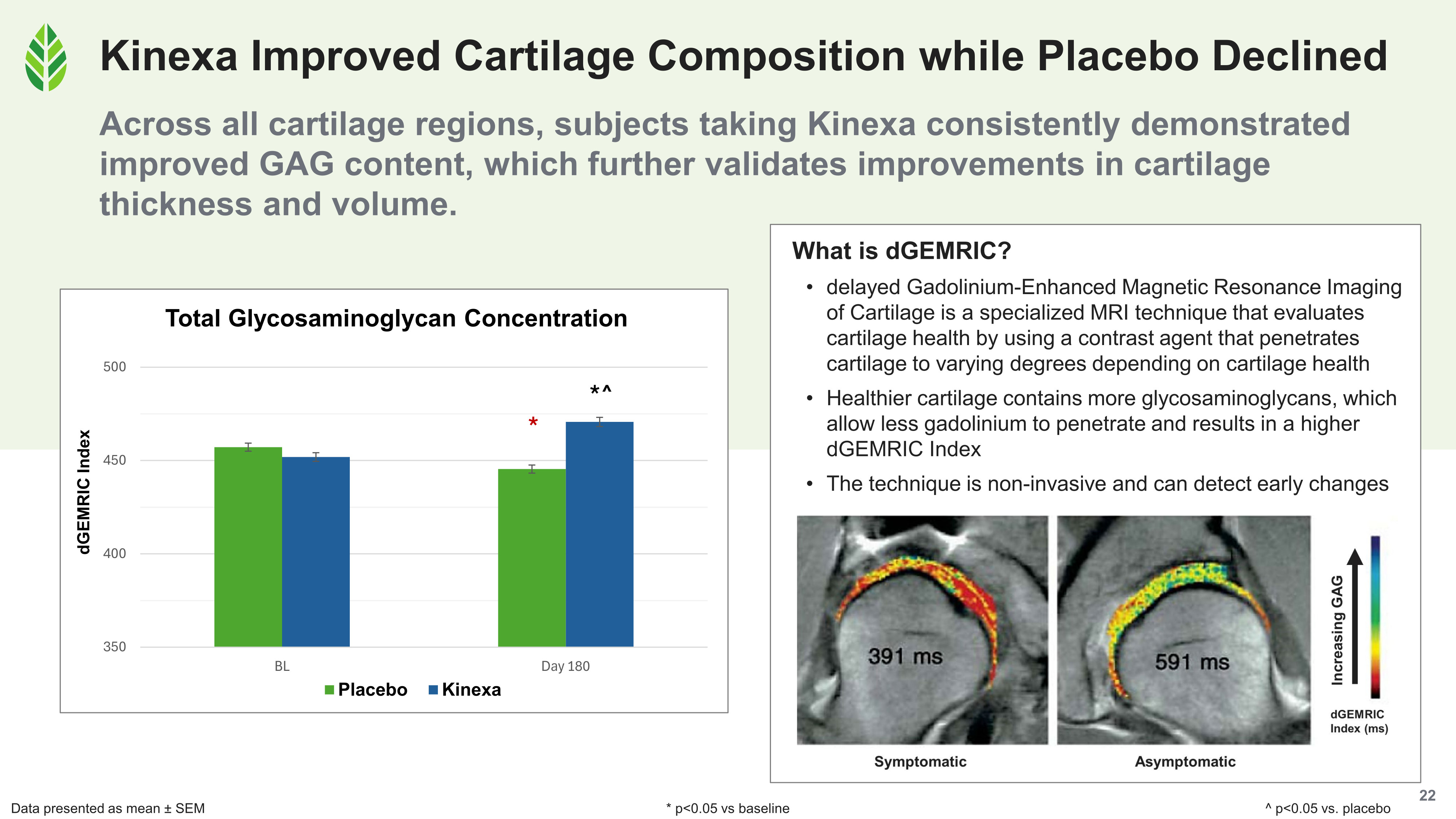
GAG concentration rose in the Kinexa group and fell in placebo subjects by day 180, confirmed by dGEMRIC imaging that directly measures cartilage biochemical quality.
Ligaments, Menisci, Synovium, and Subchondral Bone
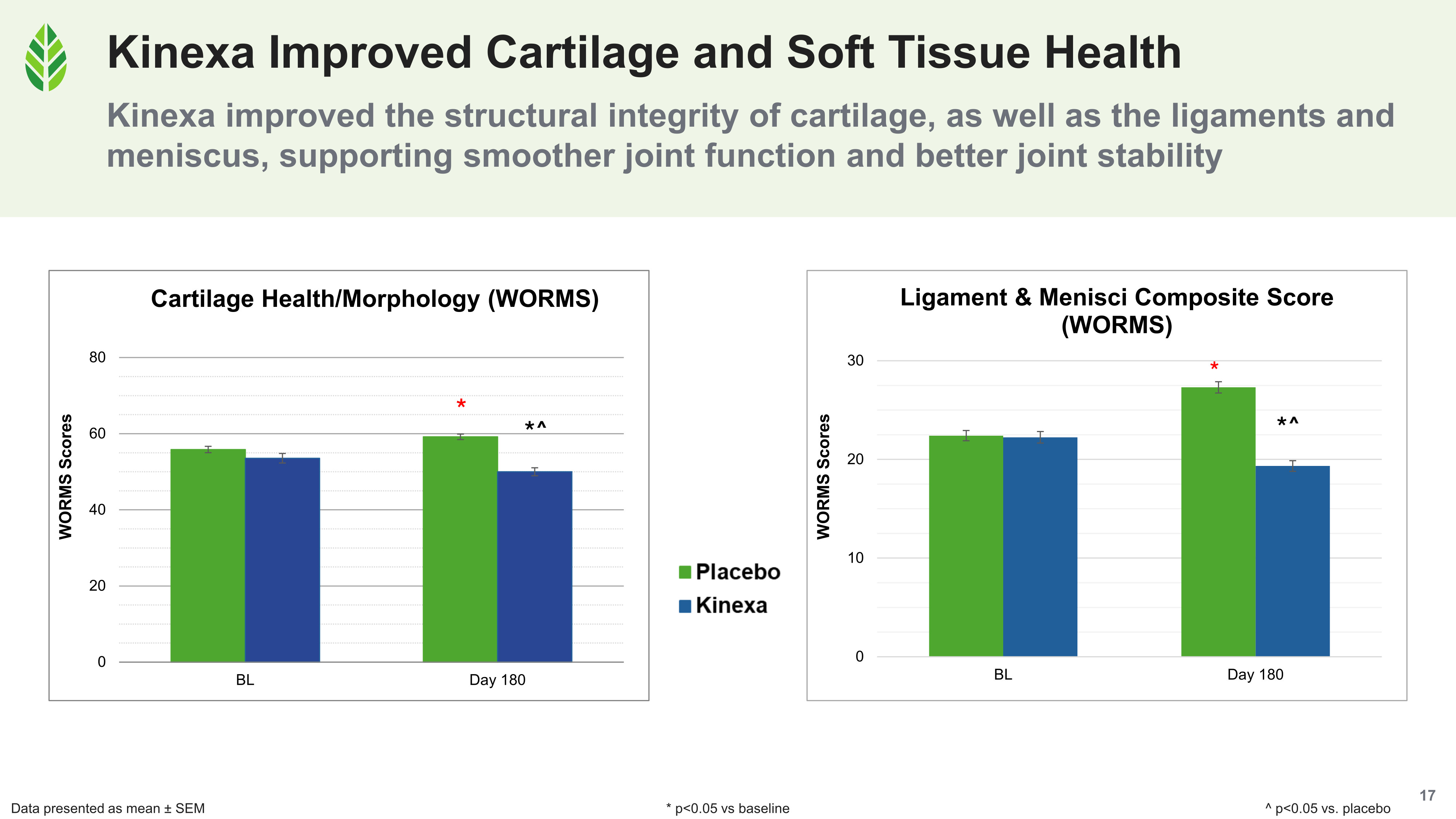
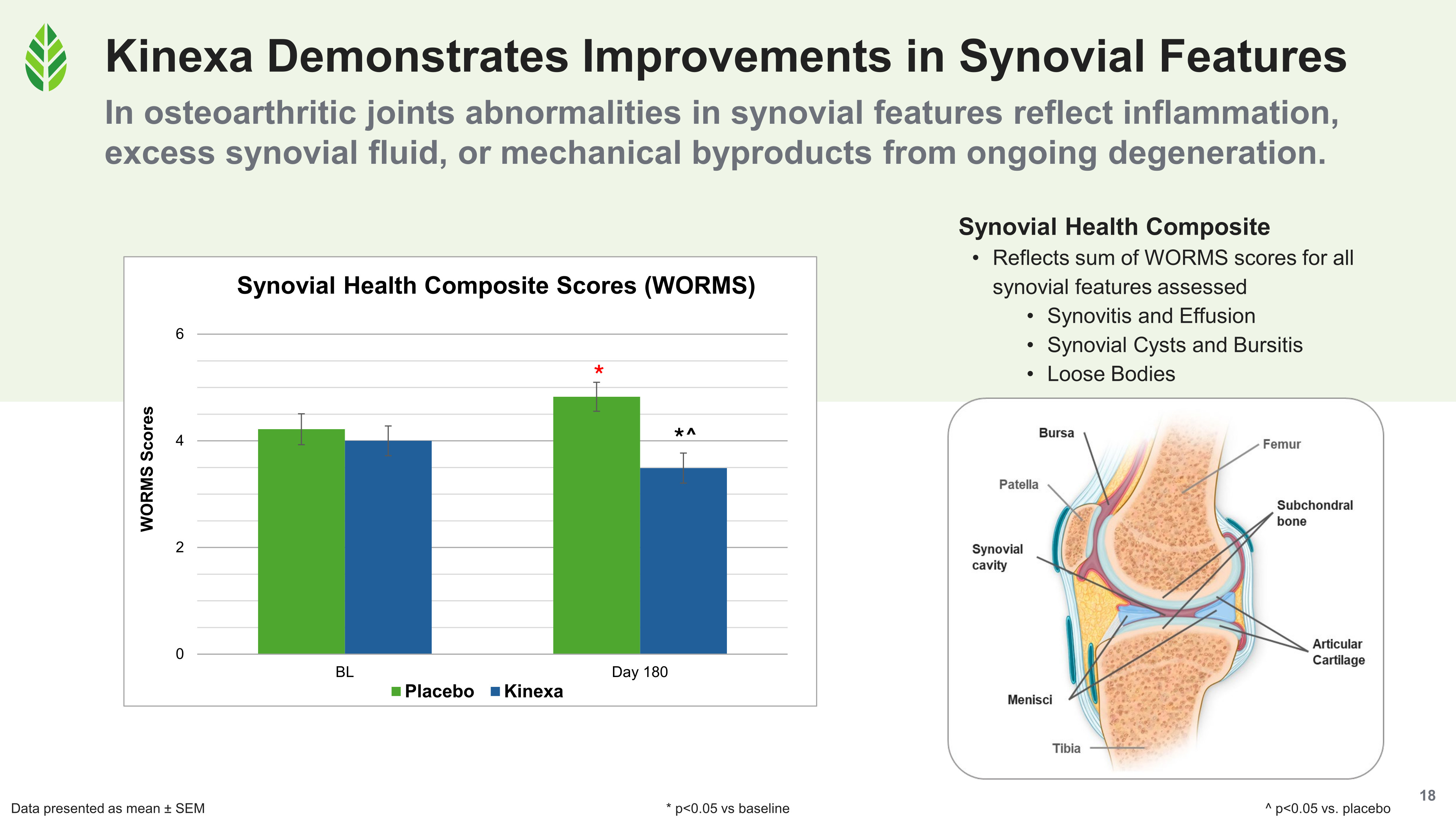
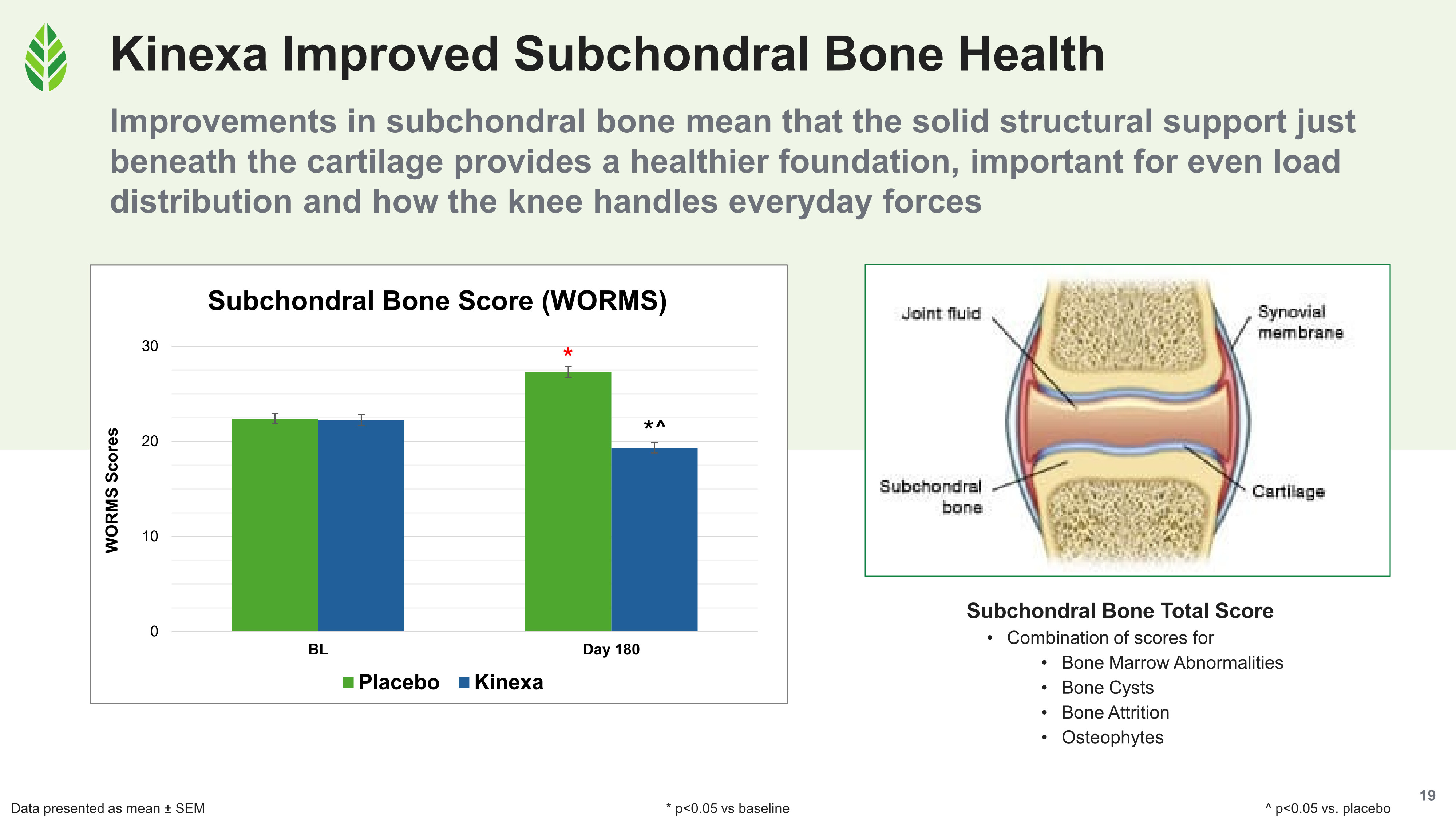
All four WORMS composite scores improved significantly versus baseline and placebo in the Kinexa group. In the placebo group, all four declined significantly over the 180-day study.
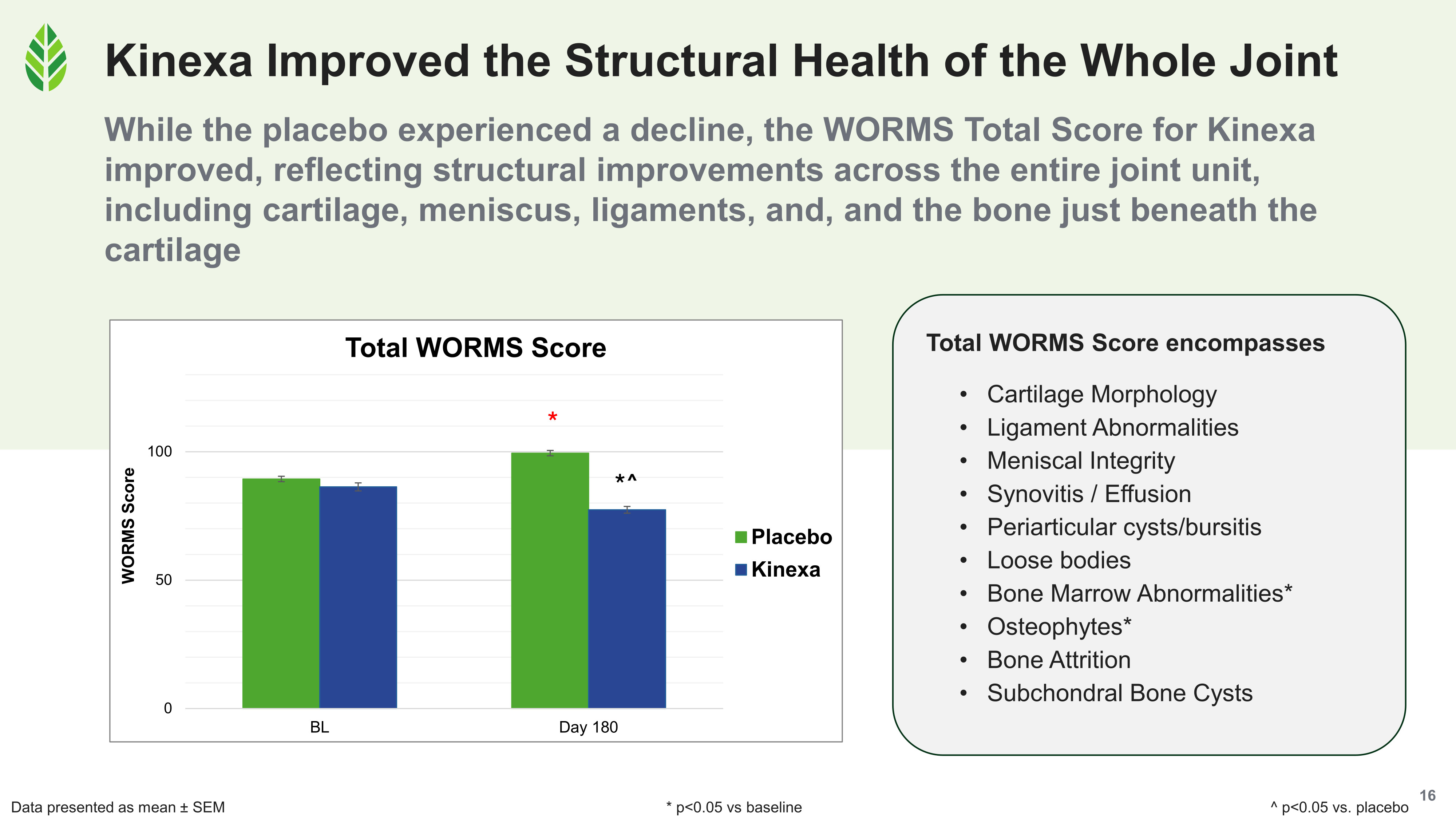
The Kinexa group's total WORMS score fell at day 180 across cartilage morphology, ligaments, menisci, synovitis, and subchondral bone, while placebo trended worse.
What does that mean practically?
- The ligaments and menisci composite (comprising anterior/posterior cruciate and medial/lateral collateral ligaments) improved in Kinexa subjects while deteriorating in controls. The menisci act as crescent-shaped shock absorbers and load distributors sitting between the femur and tibia. Their integrity is one reason certain knee injuries become more common as people age. The ligaments provide the stability that keeps the joint tracking correctly under load.
- The synovial health composite (synovitis, effusion, periarticular cysts, and loose bodies) also improved significantly with Kinexa. The synovium produces synovial fluid, which lubricates the joint and delivers nutrients to the avascular cartilage. Abnormalities here reflect ongoing inflammation and degenerative processes in the joint cavity.
- Subchondral bone composite scores improved significantly with Kinexa. This score captures bone marrow lesions, cysts, osteophytes, and bone attrition. Osteophytes (bone spurs that form along joint margins as OA progresses) matter for pain because, unlike cartilage, the surrounding soft tissue does have nerve endings. Showing that a supplement can support subchondral bone health over six months is genuinely new territory in this category.
Taken together, these five structural outcomes (cartilage, menisci, ligaments, synovium, and subchondral bone) make the Kinexa study the most structurally comprehensive clinical trial we've ever seen on a joint health dietary supplement.
Functional and Subjective Outcomes
The structural improvements translated directly into measurable functional gains, too:
- Knee strength improved an average of 24% relative to baseline in the Kinexa group by day 180 (measured by isometric knee extensor torque across 30°, 60°, and 90° angles), with significant improvements versus placebo beginning at day 30. The placebo group's torque values remained essentially flat.
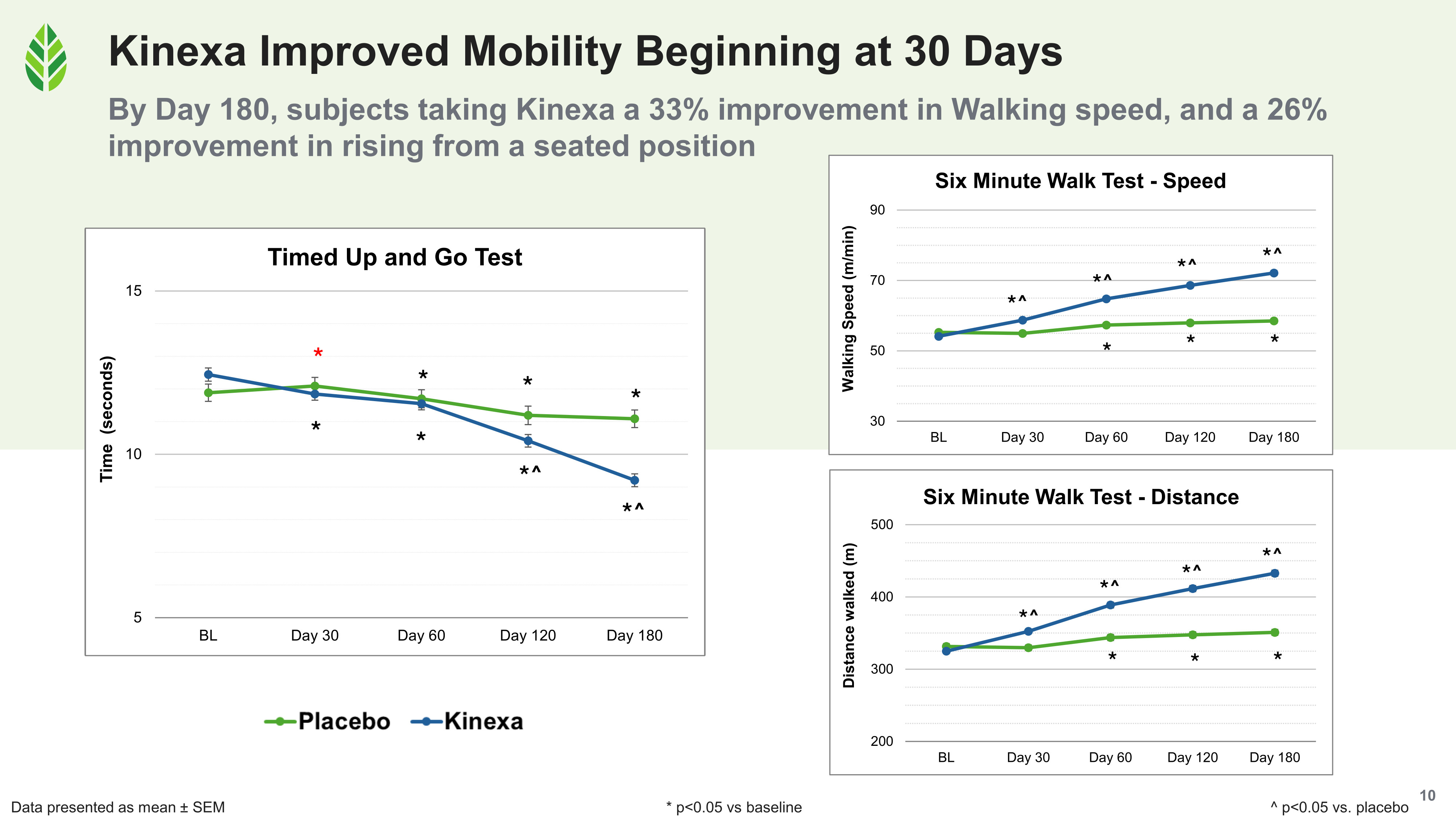
- Walking speed, measured by the Six-Minute Walk Test, improved 33% relative to baseline in Kinexa subjects. Walking distance improved correspondingly, and both were significant versus baseline and placebo at every assessment point after day 30.
- Functional mobility via the Timed Up and Go test improved 26%. This test (stand from a chair, walk a short distance, turn, walk back, sit down) is a validated assessment of mobility, balance, and fall risk. Improvements were significant versus placebo beginning at day 120 and accelerating through day 180.
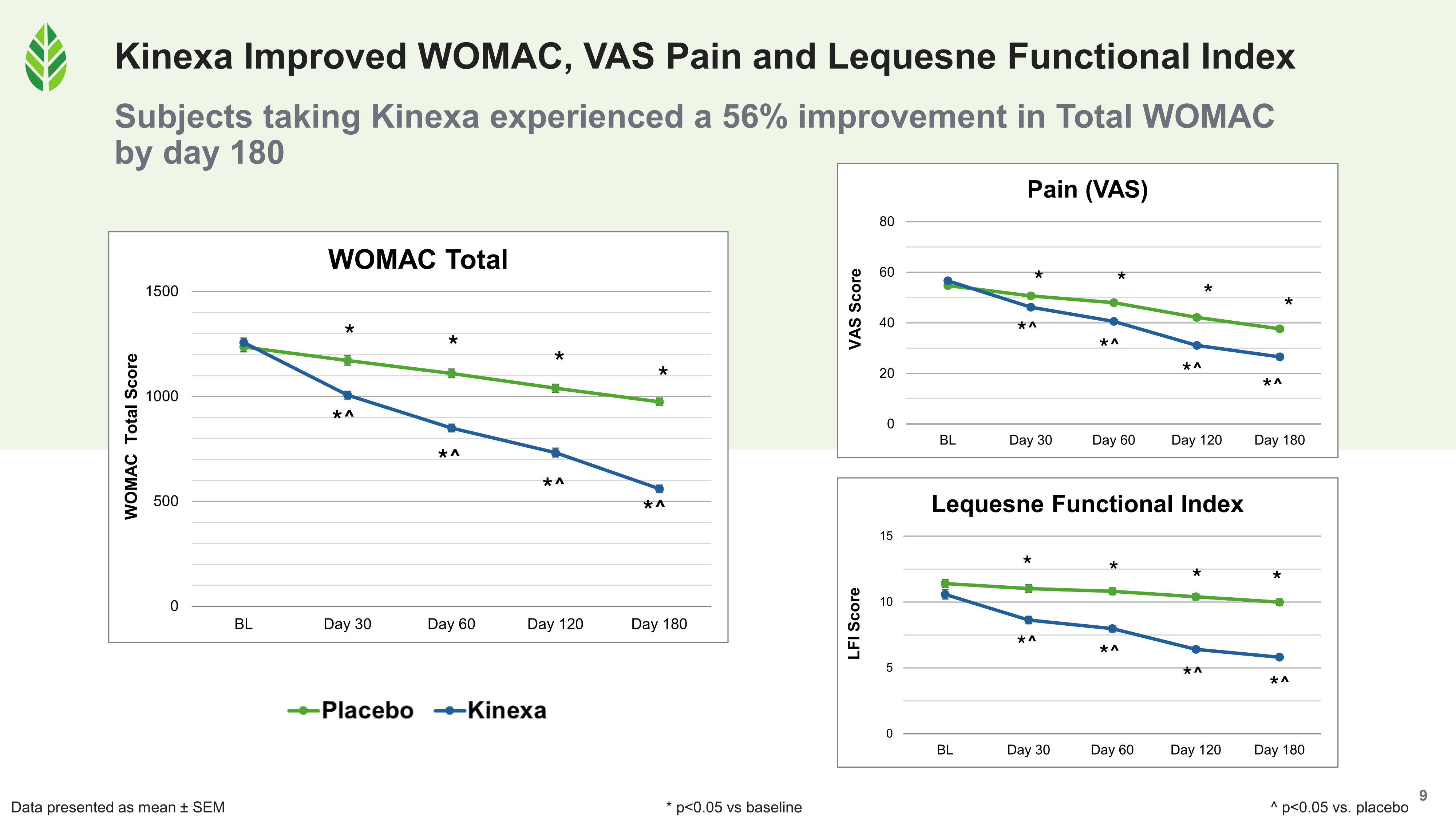
- On the subjective side, Total WOMAC improved 56% relative to baseline in the Kinexa group. Breaking that down, the pain (comfort) subset improved 60%, the stiffness subset improved 49%, and the physical function subset improved 55%, and all three subsets were significant versus baseline and placebo at every assessment from day 30 through day 180.
Between the imaging and quality-of-life scores, this is effectively earth-shattering data for the joint supplement world. And there's still more:
Biomarkers of Inflammation and Cartilage Breakdown
The study also tracked three biomarkers relevant to joint tissue health. Kinexa showed significant improvements versus baseline and placebo for hsCRP (systemic inflammation), MMP-3 (a protease involved in extracellular matrix degradation), and uCTX-II (a marker of cartilage breakdown in urine). These aren't direct structural measures, but they corroborate the MRI findings by suggesting that the processes driving cartilage degradation and inflammation slowed in the treatment group.
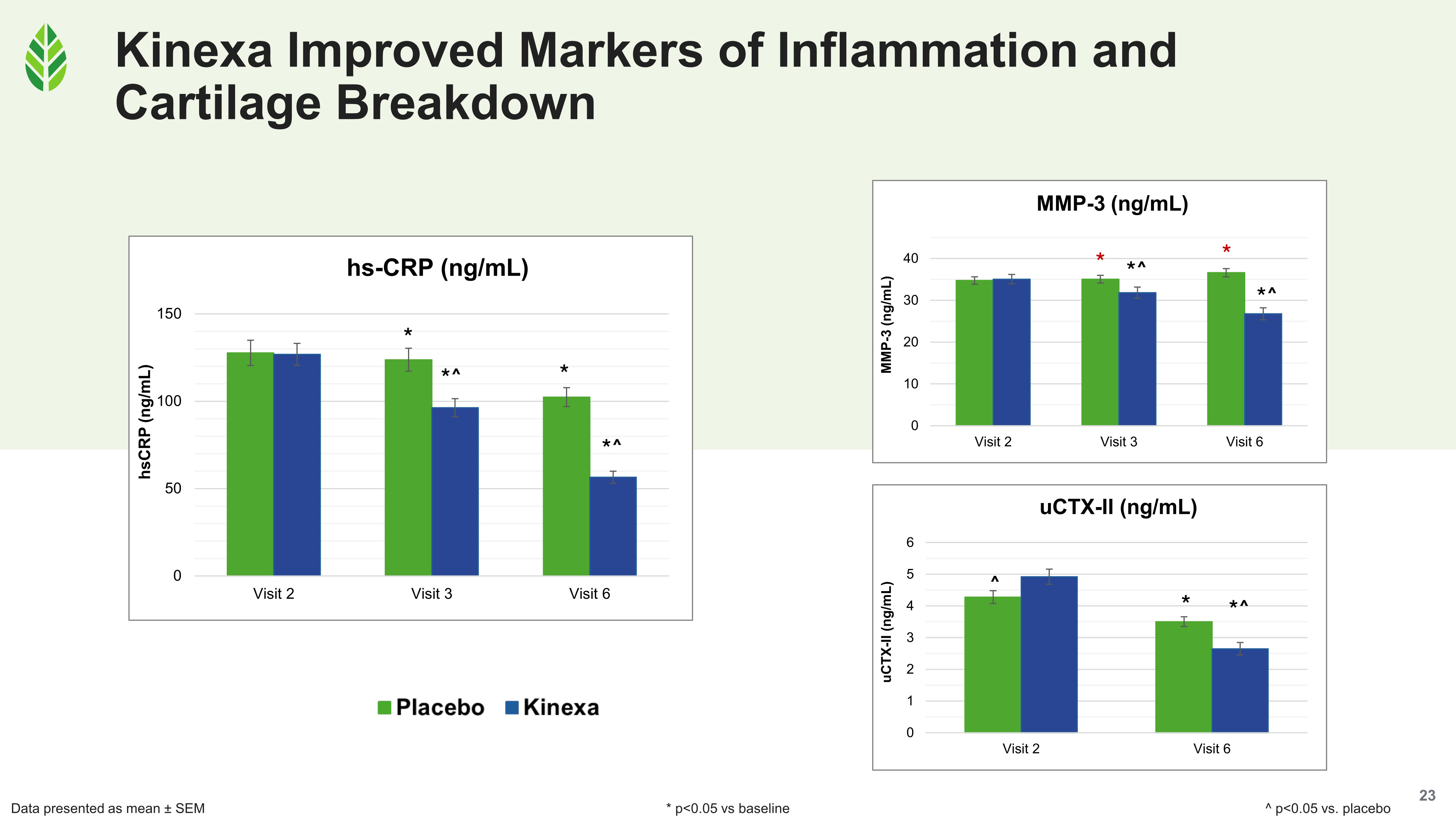
hsCRP, MMP-3, and uCTX-II all dropped significantly with Kinexa versus placebo, corroborating the MRI structural findings with biomarker-level evidence of slowed degradation.
A Brief Note on Safety
The three components of Kinexa (Terminalia chebula, Curcuma longa, and Boswellia serrata) each have well-established safety records and long histories of traditional and commercial use. All three qualify as old dietary ingredients under DSHEA. No adverse events attributable to Kinexa were reported in PLT's communications about the study. We'll update this section when the full study report is published.
Stacking and Formulation Applications
The Kinexa + AprèsFlex Stack

6-month study shows AprèsFlex delivers MRI-verified cartilage regeneration at just 100mg daily. Not just pain relief -- actual structural improvement. STROM UK's Rick Foster explains why this sets it apart from typical joint supplements.
Kinexa fills a structural longevity role that's largely absent from today's joint supplement market. But PLT also offers AprèsFlex, a specialized Boswellia serrata extract backed by four clinical studies showing fast-acting joint comfort support beginning within five days at just 100mg/day (including its own MRI-validated cartilage thickness improvements in a six-month trial).
The two ingredients work on different timelines and serve different purposes. AprèsFlex delivers experiential benefits quickly through its effects on multiple pathways, such as AKBA inhibition of 5-Lipoxygenase. Kinexa builds structural support over months, with whole-joint tissue improvements confirmed by imaging. Together, they address both immediate comfort and long-term joint integrity. PLT positions this as a natural stack for formulators building comprehensive joint products.
For context on PLT's broader Boswellia portfolio, including Dynagenix (a water-soluble, beverage-compatible Boswellia for modern delivery formats), see our PLT joint health platform overview.
Dose and Delivery Formats
The clinical and commercial dose is 200mg once daily. Kinexa is compatible with soft gels, capsules, tablets, stick packs, powders, chews, and effervescents. A Trademark License Agreement (TMLA) is required, so formulators interested in using Kinexa should contact PLT directly at PLTHealth.com.
Supplements Containing Kinexa

Dynagenix from PLT Health Solutions brings Boswellia to beverages with water solubility, neutral taste, and clinically effective doses of just 40-60mg. New research shows joint health improvements in 5 days and faster post-workout recovery.
Instaflex Complete
The first commercial product featuring Kinexa at its full 200mg clinical dose is Instaflex Complete, released by the Instaflex brand in early 2026. Each capsule delivers:
- Kinexa (Boswellia, Turmeric, Terminalia chebula extracts): 200mg
- TurmXtra 60N (Turmeric Extract, 60% curcuminoids): 250mg
- Promentum (Calcium Fructoborate): 220mg
- Collagen Peptides (bovine): 40mg
- Hyaluronic Acid: 5mg
Suggested use is one capsule daily. Instaflex positions this as their most comprehensive joint formula to date.
Instaflex Complete – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.
Joint Longevity Is Structural, and Kinexa Changes Everything

PLT Health offers four clinically-backed Boswellia extracts for joint health. AprèsFlex for capsules, Dynagenix for beverages, 5-LOXIN for high-AKBA formulas, and FLEXIR for multi-botanical support. Each optimized for different formats and consumer needs.
The joint health supplement category has done well by consumers on the comfort side of the equation. Ingredients like AprèsFlex have set a high bar for fast, clinically validated relief. But structural longevity, what's actually happening inside the cartilage, the menisci, the ligaments, the synovium, and the subchondral bone, has largely gone unmeasured. Not anymore.
Kinexa changes the questions formulators and brands can ask when building a joint product. The study is still working through the publication pipeline (expect a peer-reviewed journal article down the line; the abstract by Murphy JL, Salter D, and Kumar B is the first public output), but the clinical dataset is thorough enough that we think this ingredient has a real place in both sports nutrition and healthy aging product lines.
If you're a formulator, this one is worth looking at seriously. Even if you have a great joint formula, this is the kind of ingredient that initiates a reformulation. At 200mg once daily, it fits into a single capsule alongside a fast-acting comfort ingredient, and PLT's own data suggests the structural and subjective benefits both hold through six months of continuous use.
Stay up to date on PLT Health's ingredient developments, new research, and product launches:



Comments and Discussion (Powered by the PricePlow Forum)