Anabolic Warfare's Project Muscle has been one of the major supplement success stories in 2022, with a dozen uniquely formulated supplements designed to help its users get lean, fit, and shredded.
We've focused on the turkesterone in Project Hulk, Project Gains, and Project Bulk, as well as the resurgence of arachidonic acid in Project Jacked.

Anabolic Warfare's Project Shred takes a unique, stackable approach to dieting: through estrogen and water control!
But Project Muscle isn't all about building muscle - there are a few diet support supplements included as well. Today's is a weight loss aid that's unlike anything we've seen, because it's not your traditional thermogenic fat burner:
Project Shred: Shredding against estrogen and water
Anabolic Warfare Project Shred has no stimulants, and it doesn't even have thermogenics -- but it does work to help you achieve your shred goals through two mechanisms that often go ignored in the diet world: estrogen and water.
By including a unique and select blend of natural aromatase inhibitors and diuretics, including a few that are new to the sports nutrition industry (but not new to the world), giving you a different way to reduce "puffiness". As explained in our article below, estrogen is highly expressed in fat tissue, and can lead to a low-energy negative feedback loop.
It can all be combined with a traditional thermogenic fat burner or pre-workout supplement, but Project Shred is operating on a different -- and very stackable -- wavelength. The story's below, after showing our PricePlow-powered deals and news alerts:
Anabolic Warfare Project Shred – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.
Shredding through water and estrogen control
Project Shred is not a typical thermogenic "fat burner". Most fat burners generally have a mix of stimulants, appetite suppressants, and thermogenic spices. A good example of this is Anabolic Warfare's Phena-Lean. This is not that.
Instead, Project Shred works to help dieters get lean and reduce "puffiness" through two mechanism:
-
Estrogen control
Aromatase is the enzyme that converts anabolic hormones to estrogens (it's also known as estrogen synthase).[1] More specifically, it transforms testosterone to estradiol and androstenedione to estrone.
Too much estrogen and aromatase activity can cause "puffiness" and gynecomastia.[2,3] Numerous studies have connected estrogen production with metabolism and obesity,[4] and fat tissue contains higher levels of aromatase and estrogens,[5-7] causing a disastrous negative feedback loop.
To combat this major source of "puffiness", Project Shred has a few aromatase inhibitors inside.
-
Water control
Another source of "puffiness" is water itself. Project Shred includes a few unique diuretics, which are ingredients that increase the excretion of water, temporarily inducing weight loss.[8-10] Often, diuretics work by reducing sodium reabsorption and increasing sodium excretion,[11] and this could be a good thing if one's sodium intake or the sodium:potassium ratio is too high.
Project Shred uses a few unique diuretic ingredients -- including a few traditional ones that aren't commonplace in the sports nutrition world -- helping to slim the body down just a bit more.
It is suggested to take this supplement with water, however, and we suggest electrolyte replacement in the dosage section at the bottom.
Anabolic Warfare designed Project Shred to give dieters a different kind of edge -- because all of the thermogenic spices and fancy forms of yohimbine in the world may not do much for you if your estrogen levels are through the roof! So let's get into this unique blend:
Project Shred Ingredients
In a single three capsule serving of Project Shred from Anabolic Warfare, you get the following:
-
White Button Mushroom (Agaricus bisporus) Extract – 300 mg
Anabolic Warfare Project Shred is a powerful blend of estrogen- and water-balancing ingredients with no stimulants inside.
Anyone who wants a lean body composition, especially men, should be concerned with maintaining optimal testosterone levels.
Having low levels of testosterone leads to obesity, lack of energy, and low motivation[12] – none of which you want if you're trying to get in the gym every day, work hard to cut weight, and stay disciplined with your diet.
That's where white button mushroom extract comes in. By inhibiting aromatase,[13] the enzyme that converts testosterone to estradiol (the strongest form of mammalian estrogen), the bioactive compounds in white button mushrooms will help you keep your testosterone levels high.
This is especially important when attempting to lose weight, because long-term calorie deficits are associated with lower testosterone levels.[14]
-
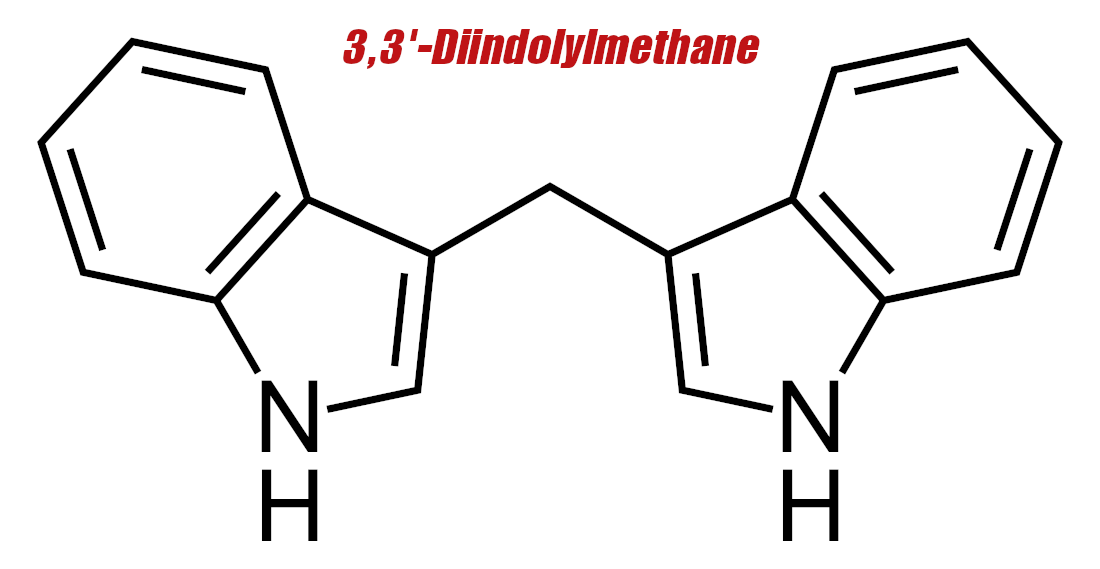
3,3'-Diindolylmethane (DIM) – 200 mg
Diindolylmethane, better known as DIM, is a phytochemical that occurs naturally in cruciferous vegetables such as broccoli, kale, and cauliflower.[15] Our gut produces DIM by metabolizing another phytochemical called indole-3-carbinol (IC3),[16,17] which anyone who's shopped around for aromatase-inhibiting supplements has probably heard of before.
Just like white button mushrooms, DIM has long been known to science as a natural aromatase inhibitor,[16] because of its affinity for the aryl hydrocarbon receptor (AhR),[18] which slightly downregulates estrogen receptors when activated.
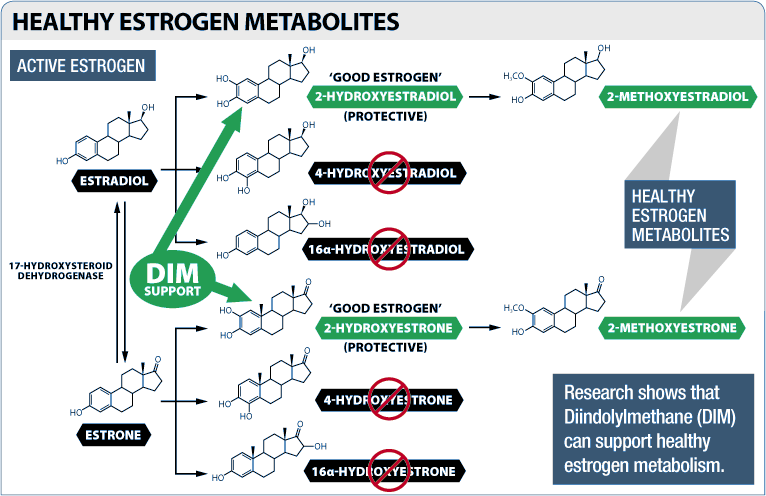
Moreover, DIM helps promote "good" forms of estrogen over the worst ones. By modulating the action of the cytochrome P450 (CYP) enzymes, DIM increases the ratio of 2-hydroxylated estrogen metabolites to 16-hydroxylated and 4-hydroxylated metabolites,[19-22] which is a good thing since 2-hydroxylated forms of estrogen have been linked to a broad array of positive health outcomes.[23,24] Conversely, the 4- and 16-hydroxylated forms are associated with negative health outcomes.
For our purposes, the thing to focus on is the fact that higher levels of 2-hydroxylated estrogens are associated with lower levels of body fat and higher levels of muscle mass.[25]
So DIM is a triple threat by:
- Increasing testosterone by inhibiting aromatase, thus...
- Decreasing estradiol levels and...
- Promoting healthy estrogen metabolites over unhealthy ones.
But that's not all - there's a broader picture to the DIM story that Project Shred also gets into:
-
Indole-3-Carbinol – 200 mg
As mentioned above, indole-3-carbinol (I3C) is the precursor to DIM.[16,17]
Although DIM, as we know, is super awesome, supplementing DIM directly is not without its pitfalls. The main concern is that, depending partly on the specific formulation of DIM, the oral bioavailability of DIM can be low.[26]
The main idea behind including I3C in this formula is that by supplying you with the precursor, which is better absorbed, you are better reaping the benefits of DIM by side-stepping any potential issues with DIM absorption in the gut.
Studies that examine I3C supplementation attest to its efficacy in fighting the proliferation of various hormone-dependent cancer cell lines,[27,28] both by decreasing their growth and also by inducing apoptosis (programmed cell death) of cancer cells.[28]
-
Buchu Leaf (Agathosma betulina) Extract – 125 mg
Used by practitioners of traditional African medicine to treat prostate inflammation and urinary tract infections,[29] buchu leaf is known to promote optimal urinary flow and kidney function[30] and is often used in supplement formulation as a diuretic.[30]
By supporting your urinary system, buchu will help you achieve the lean, shredded look you're aspiring to reach– not by increasing fat loss, but by potentially decreasing fluid retention.
-
Dandelion (Taraxacum officinale) whole plant extract – 125 mg
Dandelion is another potent herbal diuretic,[31] with a long history of use in supporting the bladder and kidneys.[31] It's considered especially safe and effective because although it has diuretic effects, it does not promote elimination of potassium from the the body.[32] This helps you maintain a healthy electrolyte balance, even as you eliminate more water from your system.
-
Juniper Berry (Juniper communis) Extract – 125 mg
Juniper berry provides significant digestive support,[33] helping you wring as much nutrition as possible from every calorie you eat while on your cut. But it's also, like dandelion and buchu leaf, a potent diuretic.[34]
Other benefits it's known for are healing the pancreas[34] and lowering blood sugar levels,[34] which, if you have weight to lose, might be a good thing since there's an association between being overweight and insulin resistance. But in some cases, juniper berry consumption can actually lower blood sugar too much,[34] so you might want to avoid this ingredient if you have a tendency for hypoglycemia.
-
Bearberry Leaf (Arctostaphylos uva-ursi) Extract – 125 mg
Similar to buchu leaf, bearberry leaf has been identified as a potential treatment for urinary tract infections,[35] and also has diuretic properties.[35]
-
Acacetin (5,7-Dihydroxy-4-methoxyflavone) – 60 mg
In one study where 24 different compounds were examined for their potential as aromatase inhibitors, acacetin was one of two that proved to have the highest anti-aromatase activity.[36] These two compounds are also known for their ability to increase libido and restore sexual function in sexually exhausted animals.[37]
-
Polygonum cuspidatum Root Powder (20% Resveratrol) – 50 mg
Also known as Japanese knotweed has long been known to traditional Chinese medicine, which uses it to improve circulation and promote cardiac health. It contains high levels of resveratrol,[38] a polyphenol that most readers of this blog have probably familiar with for its effects on the Sirtuin 1 (or SIRT1) system.
Resveratrol is also a potent aromatase inhibitor,[39] meaning that it, as we have discussed extensively already, prevents the conversion of testosterone into estrogen.
Dosage and Instructions
Project Shred is meant to be taken with 8-12oz of water. Consider replenishing electrolytes alongside your favorite electrolyte supplement.
With no stimulants inside, you can stack with a stimulant-based fat burner like Anabolic Warfare Phena-Lean or a fat-oxidation-boosting pre-workout like Stim Lord.
Conclusion: A uniquely focused supplement
Anabolic Warfare is a highly focused supplement formula – every ingredient is either an aromatase inhibitor or a diuretic.
No doubt this formula can help support fat loss, given the integral role testosterone plays in maintaining lean body mass, and the negative effect of dieting on testosterone production. But really, the orientation of this formula is more aesthetic than anything else. It will help you achieve a lean and shredded look with its emphasis on diuretics, which will significantly decrease water retention and make you appear less "puffy."
The aromatase inhibition actually has an effect on water retention as well – high estrogen levels are associated with increased water retention,[40,41] so bringing them down will have a similar aesthetic effect as the diuretics.
If you think Project Shred's take on shredding is unique -- and it is -- then you'll want to check the rest of Project Muscle. This series is loaded with similarly-strategized supplements, and you can follow it all with PricePlow:
Anabolic Warfare Project Shred – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.








Comments and Discussion (Powered by the PricePlow Forum)