Over the past two years, we've had the opportunity to highlight numerous forms of immunity-boosting ingredients such as zinc, copper, vitamin D, and even mushrooms, all of which are part of PEScience's Immune-ST formula. Today, it's time to get back to another mineral that sadly hasn't received as much attention as it deserves: magnesium.

PEScience Mag-GP introduces to a new, high-bioavailability / high-absorption form of chelated magnesium: Magnesium glycerophosphate, which they claim mixes clear, is tasteless, and non-laxative
As the second-most abundant electrolyte in our bodies, magnesium plays a critical role in bone and cell structure, metabolism, antioxidant synthesis, nerve impulses, muscle contractions, and far more.[1] Supplementing it has several benefits, as we'll cover below. However, as we've seen with various other minerals, not all forms of magnesium work the same!
PEScience Mag-GP: Magnesium Glycerophosphate Chelate
This winter, PEScience is happy to have a new magnesium alternative for you to add to your daily regimen: PEScience Mag-GP, which utilizes magnesium glycerophosphate chelate. Like the minerals used in Immune-ST, this is a chelated form of magnesium that's bound to highly absorbed molecules that provide benefits themselves.
We get into some background on Mag-GP below, and then discuss why it's so important to keep magnesium levels high on a daily basis. But first, check our coupon-powered prices on PricePlow:
PEScience Mag-GP – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.
Why Magnesium Glycerophosphate Chelate?
Dietary magnesium is never sold or packaged alone in dietary supplements - it's always going to be bound to something. We often see magnesium oxide as a low-cost form of the mineral, but just like zinc oxide, it has awfully low bioavailability - as low as 4%.[2]
Meanwhile, magnesium found in food generally has a 20-30% absorption rate,[3-5] although some leafy vegetables can range as high as 40-60%.[6,7] In general, absorption is roughly one-third, in line with what we see from magnesium citrate,[8,9] a more common form.
Using mineral glycerophosphates (combining glycerol, phosphate, and magnesium)
In an attempt to do even better, PEScience has opted for magnesium glycerophosphate, which is made using glycerophosphoric acid (also known as glycerol-1-phosphate), which takes glycerol (found in PEScience's High Volume pre workout) bound to a phosphate group.[10]
These are both beneficial in their own right, and the idea is to attach magnesium[11] (something that isn't absorbed well) to glycerol (something that is absorbed well). The kicker is that the phosphate plays a parallel role with magnesium, as it's needed for the production of our cellular "energy currency" adenosine triphosphate, also known as ATP.[12]
There isn't much peer-reviewed research on magnesium glycerophosphate itself, but it's interesting to note that when researchers need a very high-quality form of the mineral (such as in situations with cardiac disease), this is one that they go to.[13]
As a sugar alcohol, glycerol has a positive taste and solubility, and this leads to PEScience's advertised benefits:
- "Taste-free"
- "Dissolves easily"
- "Digests perfectly"
- "Non-laxative"
Mag-GP Dosage
As commonly seen with other chelated minerals, it takes a good amount of powder to get an efficacious dose -- Mag-GP's content is around 12% magnesium by weight, so you'll need roughly 3.5 grams total if you want to get 420 milligrams of magnesium, or ~100% daily recommended intake.
Generally, you'll get some magnesium from your diet so it's important to understand what you eat and how much you need to save on money.
Why supplement magnesium?
With so many biochemical reactions that require magnesium, it's incredibly important to sustain high enough levels. Deficiency can lead to numerous conditions, including anxiety, muscle weakness, hypertension, and mood disorders.[14] However, magnesium deficiency is very difficult to diagnose, since serum magnesium levels may seem fine because the body is pulling magnesium from bone tissue and cells where it's needed.[14-16] Making matters worse, symptoms can range widely amongst individuals.[17,18]
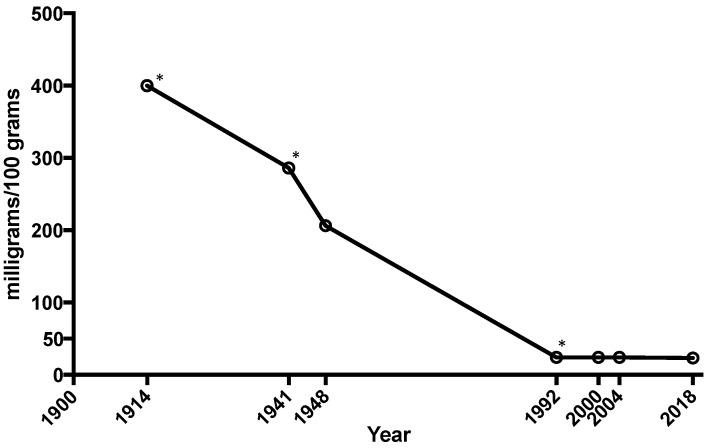
The average amount of calcium, magnesium, and iron in vegetables like spinach, lettuce, cabbage, and tomatoes has plummeted as much as 80–90% since 1914.[21] Sadly we have to supplement it back in.
The average American is not getting anywhere near the recommended amount of magnesium,[19,20] and given the drastically reduced amount of magnesium content in our food thanks to poor farming practices and high time preference soil management,[21-27] we're led to believe that Westerners are generally running a "low-level" magnesium deficiency at best, and moderate deficiencies is more likely.
Nearly everyone should supplement at least some
The solution -- until we fix our soil and food production through regenerative grazing and agriculture -- is to universally supplement it, finding a dose that feels good but doesn't provide side effects. This will generally be between 50-100% DV, or somewhere between one and two scoops of Mag-GP.
Magnesium Benefits in human research
The following benefits have been demonstrated in humans, but note that they were done using alternative forms of magnesium. So long as we have a bioavailable form, we believe it's OK to cite them in this article:
- Better sleep[28,29]
- Improved mood / reduced anxiety[30,31]
- Lower blood pressure[32-35]
- Reduced HbA1c and blood glucose levels[32,36,37]
- Decreased fasting insulin levels[32,36,38]
- Improved insulin sensitivity[32,36,37]
- Better bone mineral density[39]
- Lower PMS symptoms[30,40-42]
The benefits go on, and given the metabolic and cardiac improvements, we feel that it's a great idea to supplement magnesium, and with the taste and solubility benefits of PEScience Mag-GP, this is one that you can easily add to any drink for some incredible benefits.
If you can add it in a small amount of water, it could be worth considering before bed, as many have noted sleep improvements when using it in this fashion.
A new magnesium worth trying
PEScience has brought us many innovative supplements over the years, but it's always great to get back to the foundational basics of improved mineral status. Just like Immune-ST brought us chelated minerals we now have a new form of magnesium to test. Whether it's for bone strength, better sleep or improved metabolism, the magnesium glycerophosphate in Mag-GP is one absolutely worth trying for anyone on their health and wellness journey.
PEScience Mag-GP – Deals and Price Drop Alerts
Get Price Alerts
No spam, no scams.
Disclosure: PricePlow relies on pricing from stores with which we have a business relationship. We work hard to keep pricing current, but you may find a better offer.
Posts are sponsored in part by the retailers and/or brands listed on this page.





Comments and Discussion (Powered by the PricePlow Forum)